Research Article
Variation in Germination and Growth Rates of Two Isolates of Beauvaria bassiana (Balsamo) Vuillemin (Deuteromycota: Hyphomycetes at Different Temperatures and their Virulence to Callosobruchus maculatus (Fabricius) (Coleoptera: Bruchidae)
Department of Life Sciences, University of the West Indies, St. Augustine Trinidad, West Indies
Ayub Khan
Department of Life Sciences, University of the West Indies, St. Augustine Trinidad, West Indies









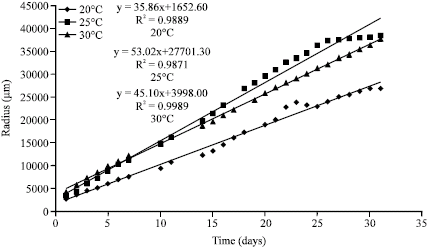
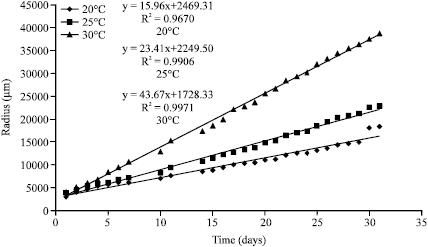

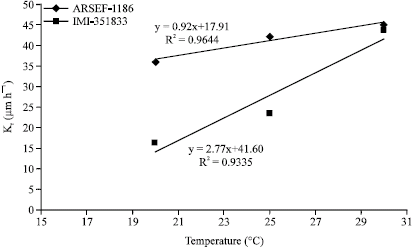
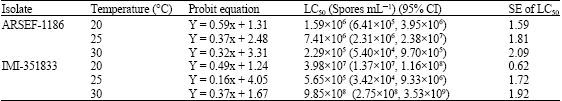
Jeff Lord Reply
Beauveria is spelled incorrectly in this paper.