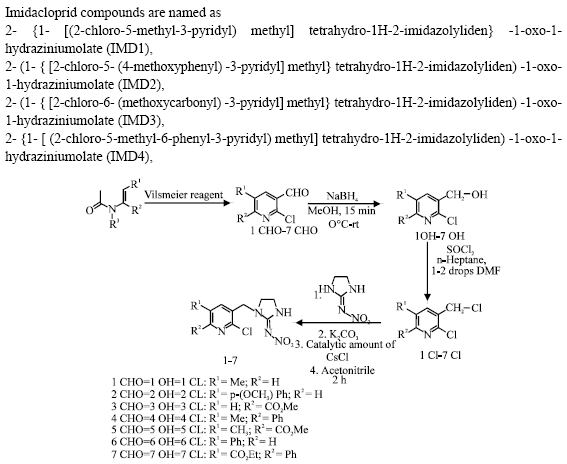
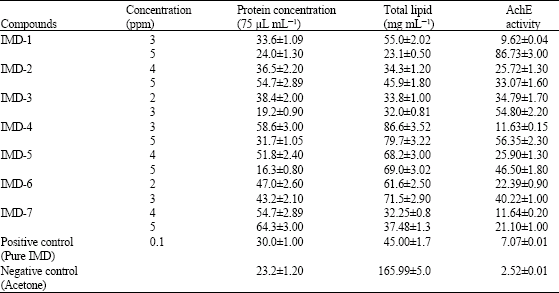
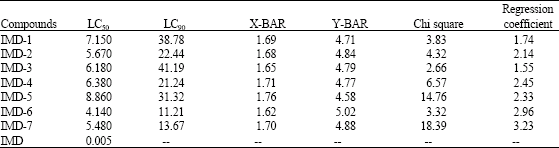Research Article
Larvicidal Efficacy of Neonicotinoid Classes of Compounds on Culex quinquefasciatus
Biology Division, Indian Institute of Chemical Technology, Uppal Road, Tamaka, Hyderabad 500 007, India
U.S. N. Murty
Biology Division, Indian Institute of Chemical Technology, Uppal Road, Tamaka, Hyderabad 500 007, India
B. Gangadasu
0rganic Chemistry Division-Il, Indian Institute of Chemical Technology, Uppal Road, Tamaka, Hyderabad 500 007, India
B. China Raju
0rganic Chemistry Division-Il, Indian Institute of Chemical Technology, Uppal Road, Tamaka, Hyderabad 500 007, India
C.H. Ramesh
0rganic Chemistry Division-Il, Indian Institute of Chemical Technology, Uppal Road, Tamaka, Hyderabad 500 007, India
S. Bharat Kumar
0rganic Chemistry Division-Il, Indian Institute of Chemical Technology, Uppal Road, Tamaka, Hyderabad 500 007, India
V. Jayathirtha Rao
0rganic Chemistry Division-Il, Indian Institute of Chemical Technology, Uppal Road, Tamaka, Hyderabad 500 007, India