Research Article
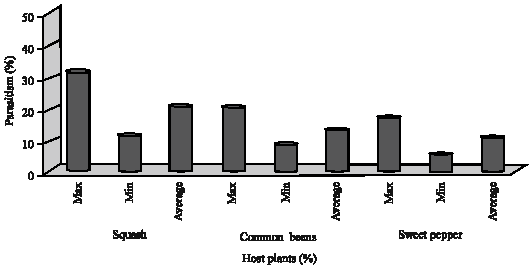
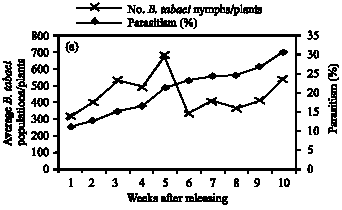
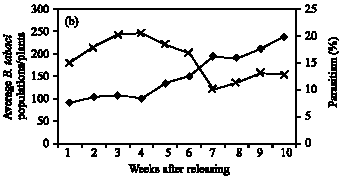
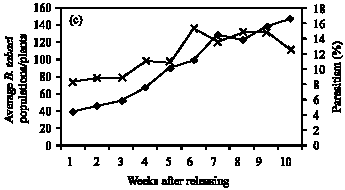
Role of Host Plants on the Biological Aspects and Parasitism Levels of Eretmocerus mundus Mercet (Hymenoptera: Aphelinidae), a Parasitoid of Bemisia tabaci (Gennadius) (Hemiptera: Aleyrodidae)
Department of Crop Production and Protection, College of Agriculture and Veterinary Medicine, Qassim University, P.O. Box 6622, Buraydah 51452, Kingdom of Saudi Arabia
M.A. Al-Deghairi
Department of Crop Production and Protection, College of Agriculture and Veterinary Medicine, Qassim University, P.O. Box 6622, Buraydah 51452, Kingdom of Saudi Arabia