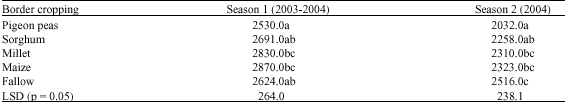
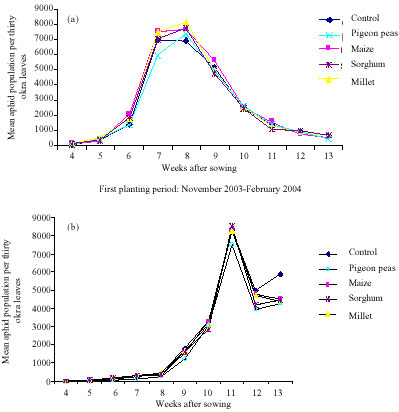
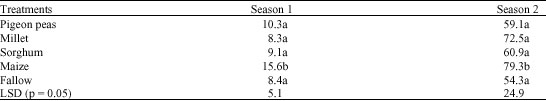
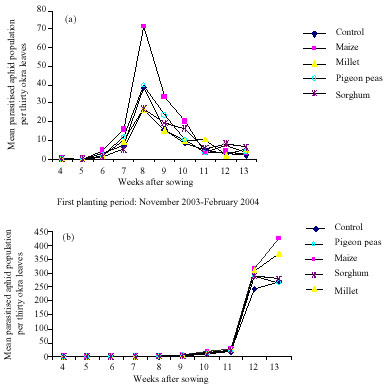
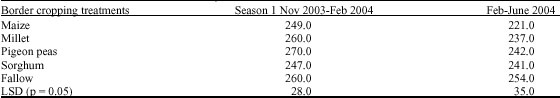
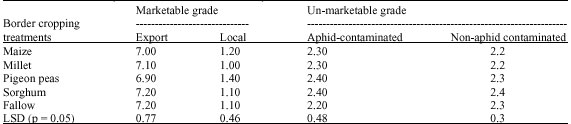
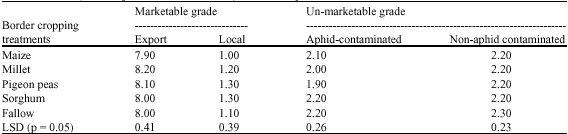
Research Article
Evaluating Border Cropping System for Management of Aphids (Hemiptera: Aphididae) Infesting Okra (Malvaceae) in Kenya
Department of Plant Science and Crop Protection, University of Nairobi, P.O. Box 30197-00100, Nairobi, Kenya
M. Kasina
National Agricultural Research Laboratories, Kenya Agricultural Research Institute,P.O. Box 14733-00800, Nairobi, Kenya
F. Malenge
Department of Plant Science and Crop Protection, University of Nairobi, P.O. Box 30197-00100, Nairobi, Kenya