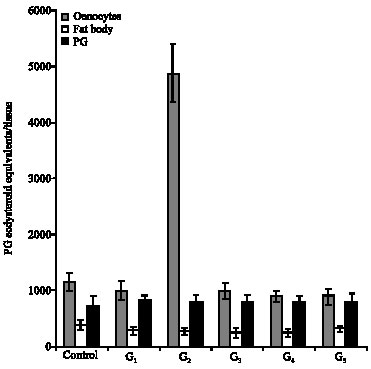
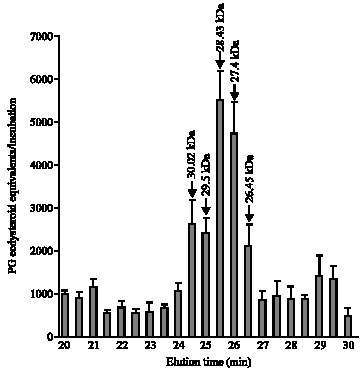
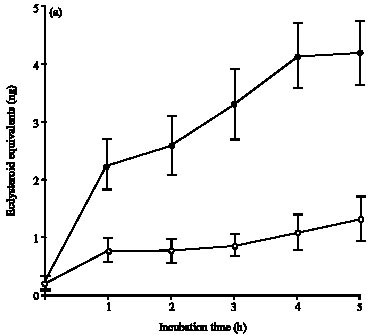
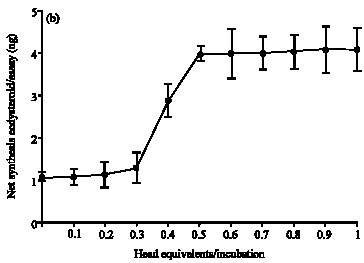
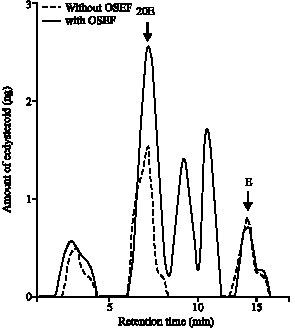
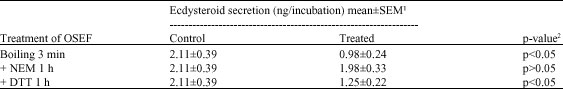
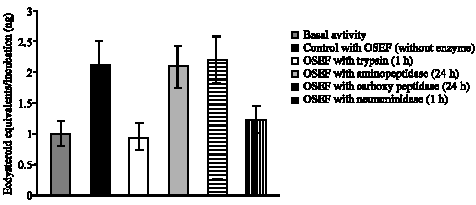
Research Article
A New Ecdysiotropic Factor in Adult Male Crickets
Department of Biology, Johannes Gutenberg University of Mainz, D-55099 Mainz, Germany
Franz Romer
Department of Biology, Johannes Gutenberg University of Mainz, D-55099 Mainz, Germany