Research Article
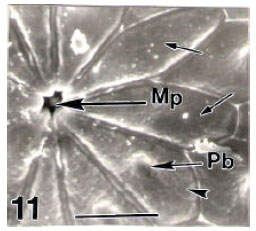
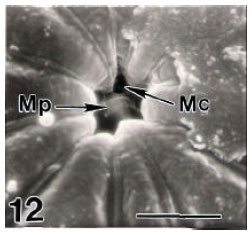
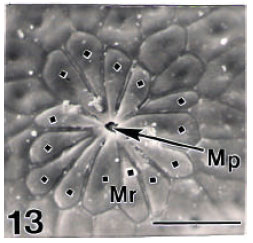
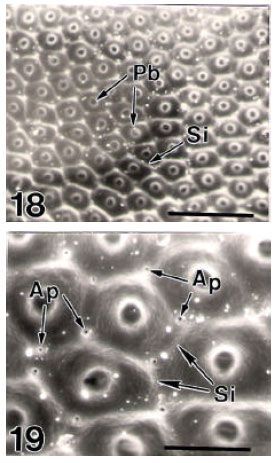
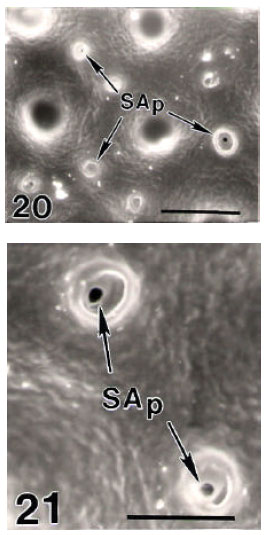
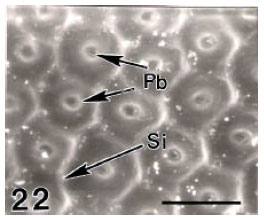
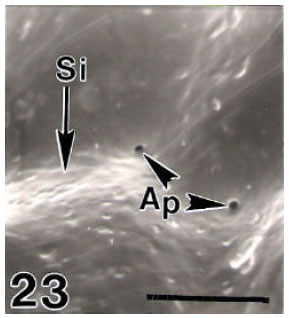
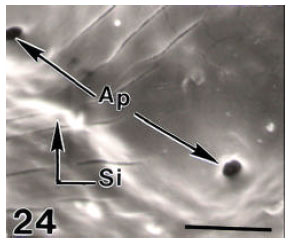
Surface Ultrastructure of the Egg Chorion of Eri Silkworm, Samia ricini (Donovan) (Lepidoptera: Saturniidae)
Electron Microscopy Division, India
B.K. Kariappa
Central Sericultural Research and Training Institute, Srirampura, Mysore-570 008, India
A.M. Babu
Electron Microscopy Division, India
S.B. Dandin
Central Sericultural Research and Training Institute, Srirampura, Mysore-570 008, India









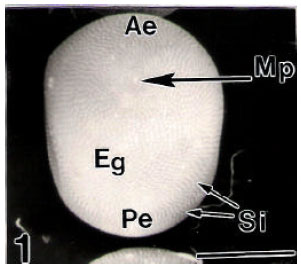
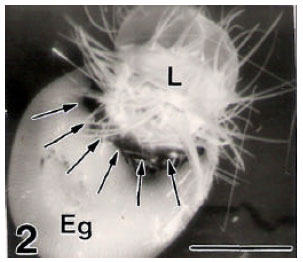
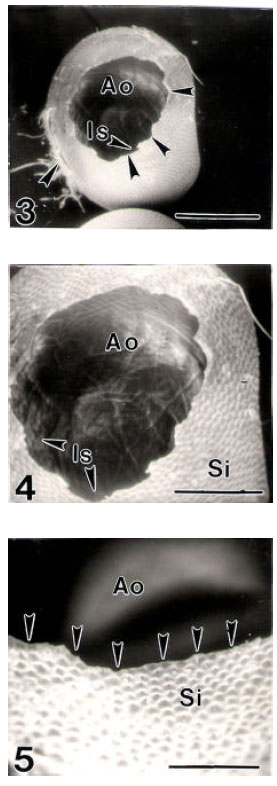
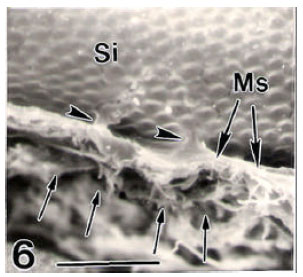



ASUTOSH KUMAR SRIVASTAVA Reply
Respected sirs,
an excellant work through SEM. As I am doing my Doctorate in Insect Endocrinology it become very use full for me.
i want to become in regular regular touch with you (all) sir so kindly forward e-mail id of yours.