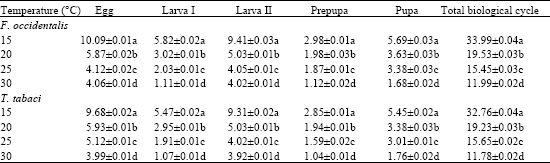
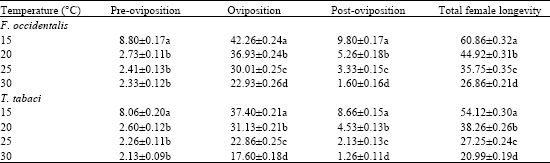
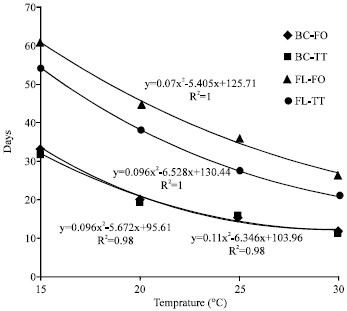
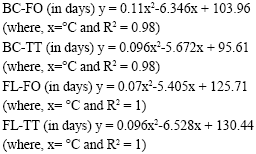Research Article
Longevity and Reproduction of Frankliniella occidentalis and Thrips tabaci on Cucumber under Controlled Conditions
Institute of Western Macedonia, Branch of Florina, Department of Crop Production, Terma Kontopoulou, 53100 Florina, Greece
L. Giakalis
Institute of Western Macedonia, Branch of Florina, Department of Crop Production, Terma Kontopoulou, 53100 Florina, Greece
G. Sidiropoulos
Institute of Western Macedonia, Branch of Florina, Department of Crop Production, Terma Kontopoulou, 53100 Florina, Greece
M. Vaiopoulou
Institute of Western Macedonia, Branch of Florina, Department of Crop Production, Terma Kontopoulou, 53100 Florina, Greece
G. Kaltsoudas
Institute of Western Macedonia, Branch of Florina, Department of Crop Production, Terma Kontopoulou, 53100 Florina, Greece
C. G. Ipsilandis
Institute of Western Macedonia, Branch of Florina, Department of Crop Production, Terma Kontopoulou, 53100 Florina, Greece