Research Article
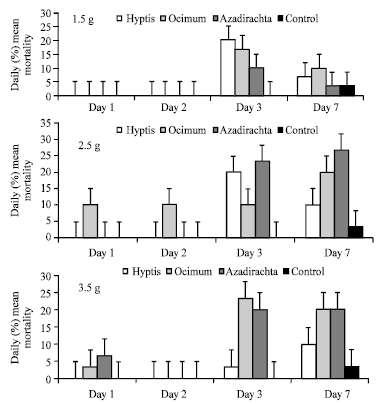
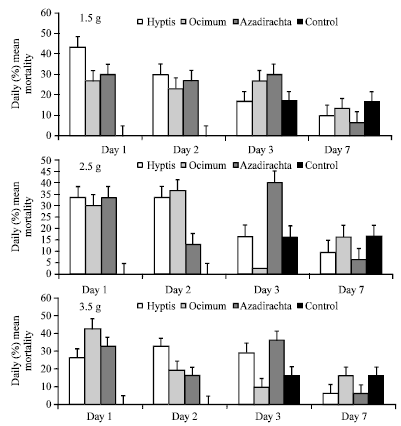
Daily Mortality Responses of Callosobruchus maculatus and Sitophilus zeamais to Changes in the Concentrations of Azadirachta indica, Ocimum gratissimum and Hyptis suaveolens
Department of Animal and Environmental Biology, University of Benin, Benin City, Nigeria
T. Ekrakene
Department of Basic Sciences, Benson Idahosa University, Benin City, Nigeria