Research Article
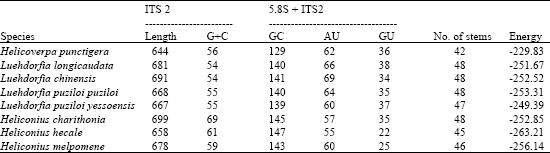
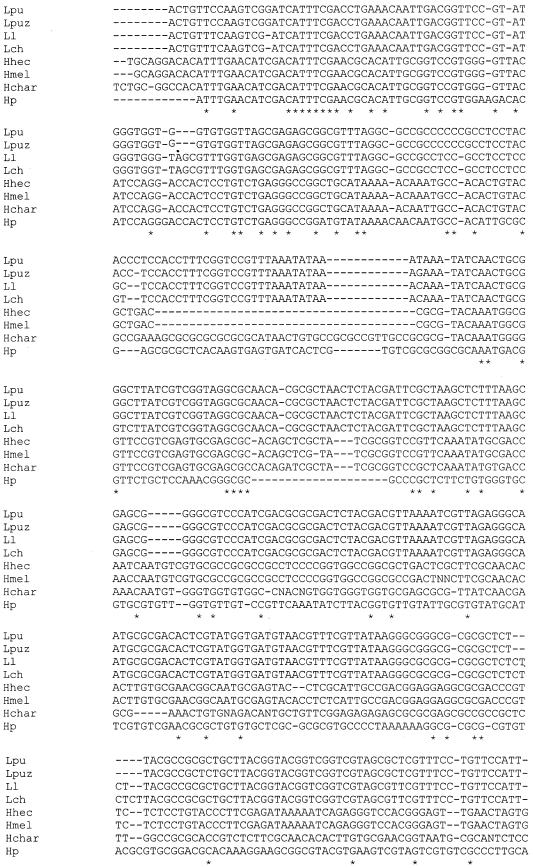
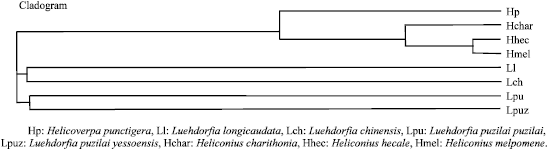
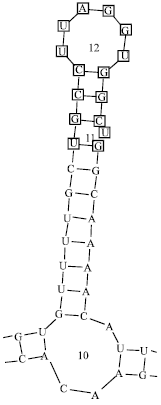
Comparison of Secondary Structure of the Ribosomal Internal Transcribed Spacer 2 of Eight Lepidopteran species from Diverse Geographical Locations
Department of Zoology, Osmania University College for Women, Koti, Hyderabad, Andhra Pradesh, India
Bhargavi Rayavarapu
Department of Zoology, Osmania University College for Women, Koti, Hyderabad, Andhra Pradesh, India
Venkata Rama Satya Kumar Duvvuri
Department of Epidemiology and Preventive Medicine, University of Maryland, Baltimore, Maryland, USA
Poduri Nagaraja Rao
Department of Zoology, Osmania University College for Women, Koti, Hyderabad, Andhra Pradesh, India