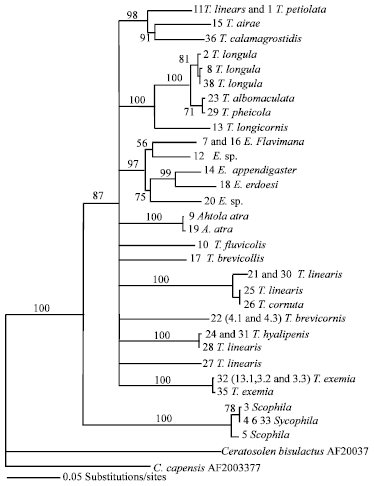
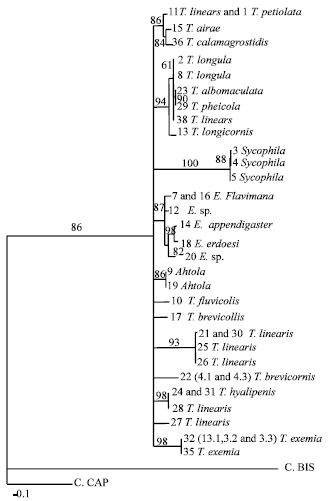
Research Article
Mitochondrial Phylogenetics of UK Eurytomids
Department of Plant Protection, Aburayhan Compas, University of Tehran Tehran P.O. Box, Iran
Mike Bruford
Department of Ecology, School of Bioscience, Cardiff University, UK
Hassan
Department of Biology, King Khalid University, Saudi Arabia
LiveDNA: A. Dawah
Carlos
Department of Ecology, School of Bioscience, Cardiff University, UK
LiveDNA: Fernandesand Ciara S. Dodd