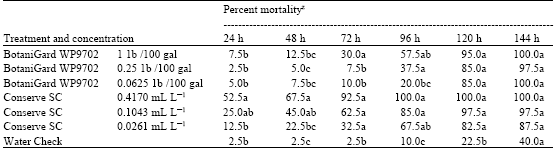
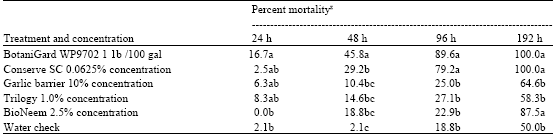
Research Article
Evaluation of Microbial and Repellent Insecticides for Control of Migratory Grasshopper, Melanoplus sanguinipes (Fabricius), in Colorado
Department of Plant Protection, Faculty of Agriculture, MustafaKemal University, 31034, Antakya, Ratay, Turkey
W. Cranshaw
Department of Bioagricultural Sciences and Pest Management, Colorado State University, Ft. Collins, CO 80523, USA