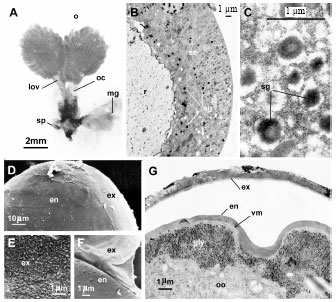
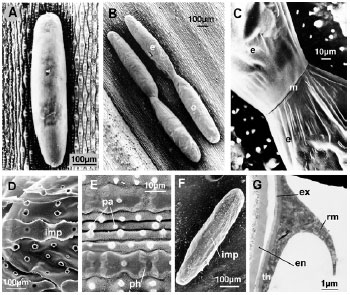
Research Article
Anatomy of the Female Reproductive System and the Ultrastructure of the Egg’s Envelopes of Orseolia oryzivora (Diptera: Cecidomyiidae)
Campus de Beaulieu, Faculte des Sciences, Universite de Rennes I, Avenue du General Leclerc, F-34042 Rennes Cedex France
Jean-Pierre Nenon
Campus de Beaulieu, Faculte des Sciences, Universite de Rennes I, Avenue du General Leclerc, F-34042 Rennes Cedex France