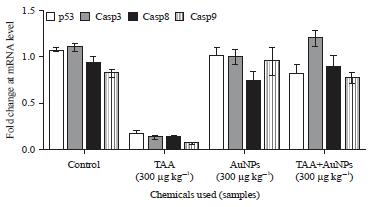
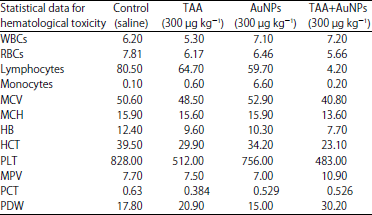
Research Article
Molecular Investigation of Gold Nanoparticles Toxicity in Mice Model and p53 Activation
Department of Biotechnology, Virtual University of Pakistan, Lahore, Pakistan
Mehmet Ozaslan
Deparment of Biology, University of Gaziantep, Gaziantep, Turkey
LiveDNA: 90.581
Hafiz Khawar Ali
Department of Molecular Biology, Virtual University of Pakistan, Lahore, Pakistan
Muhammad Safdar
Department of Breeding and Genetics, Cholistan University of Veterinary and Animal Sciences, 63100 Bahawalpur, Pakistan
LiveDNA: 92.25537
Yasmeen Junejo
Department of Physiology and Biochemistry, Cholistan University of Veterinary and Animal Sciences, 63100 Bahawalpur, Pakistan
Masroor E. Babar
Department of Molecular Biology, Virtual University of Pakistan, Lahore, Pakistan