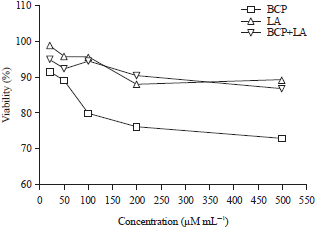
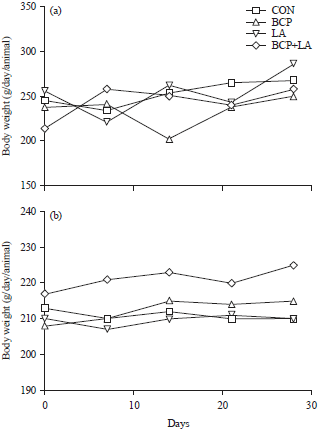
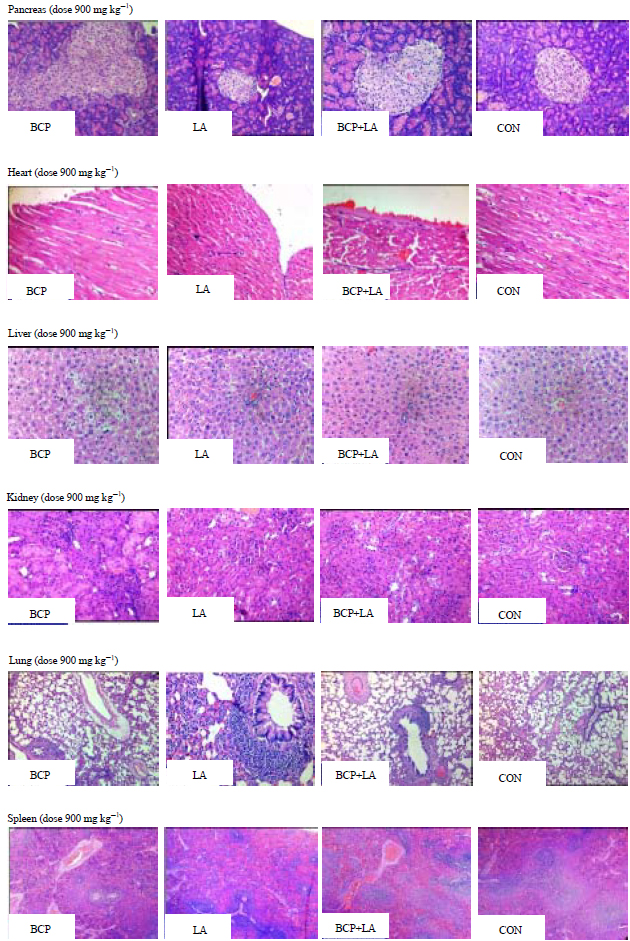
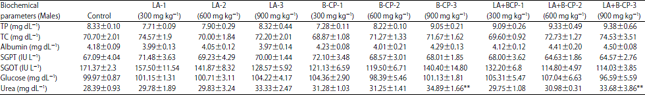
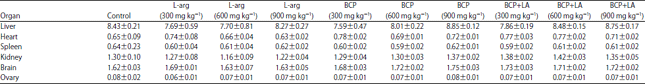
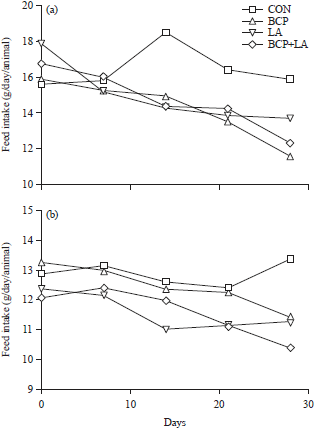
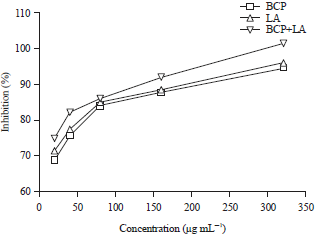
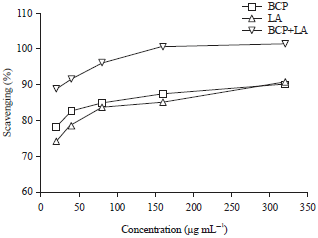
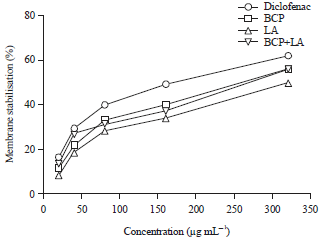
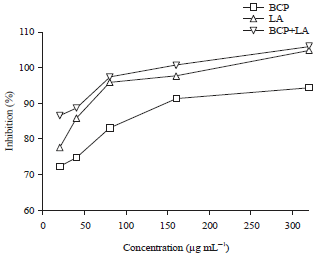
Research Article
Evaluation of Safety and in vitro Mechanisms of Anti-diabetic Activity of β-caryophyllene and L-arginine
Shobhaben Pratapbhai Patel School of Pharmacy and Technology Management, SVKM`s NMIMS, V.L. Mehta Road, Vile Parle (W), 400056 Mumbai, India
LiveDNA: 91.3069
Leo Joseph Philip Tharappel
Shobhaben Pratapbhai Patel School of Pharmacy and Technology Management, SVKM`s NMIMS, V.L. Mehta Road, Vile Parle (W), 400056 Mumbai, India
Vivek Kumawat
Shobhaben Pratapbhai Patel School of Pharmacy and Technology Management, SVKM`s NMIMS, V.L. Mehta Road, Vile Parle (W), 400056 Mumbai, India