Research Article
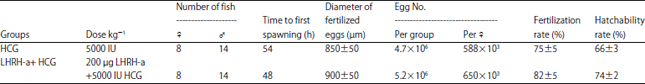
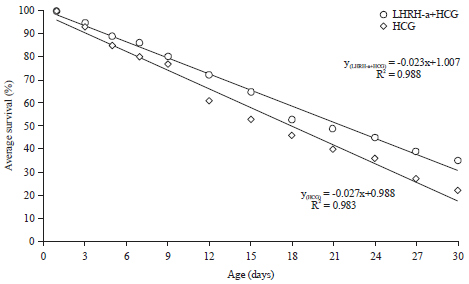
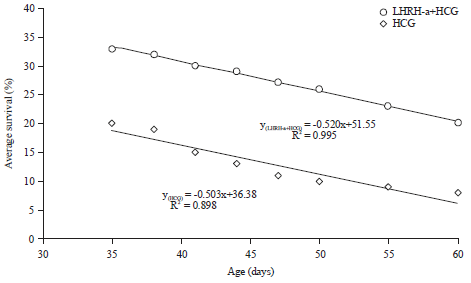
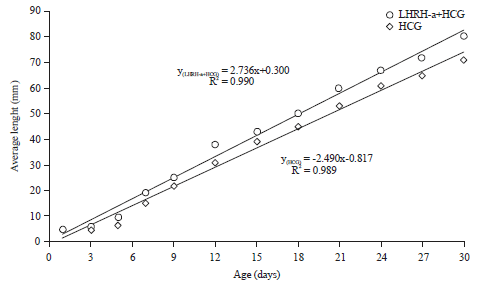
Reproductive Performance of Sea Bream (Sparus aurata) Induced with Two Different Hormone Protocols with Respect to the Effect on Gonadotrophic Cells
Department of Fish Reproduction and Aquacultural, National Institute of Oceanography and Fisheries, Aquaculture Division, El-Anfousy, Kayetbay, Alexandria, Egypt
LiveDNA: 20.16715
Zeinab Abdel Baki El-Greisy
Department of Fish Reproduction and Aquacultural, National Institute of Oceanography and Fisheries, Aquaculture Division, El-Anfousy, Kayetbay, Alexandria, Egypt
LiveDNA: 20.19539
Heba Saad El-Sayed
Department of Fish Reproduction and Aquacultural, National Institute of Oceanography and Fisheries, Aquaculture Division, El-Anfousy, Kayetbay, Alexandria, Egypt
LiveDNA: 20.16883