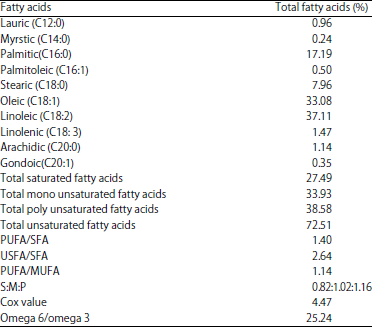
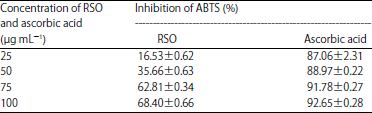
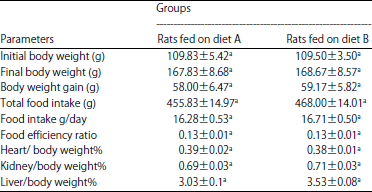
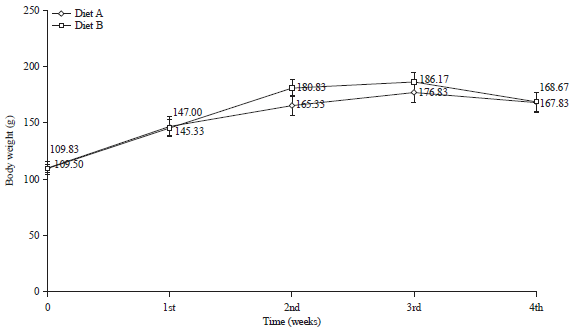
Research Article
Roselle Seed as a Potential New Source of Healthy Edible Oil
Department of Nutrition and Food Sciences, National Research Centre, Dokki, Cairo, Egypt
LiveDNA: 20.16948
Adel Gabr Abdel-Razek
Department of Fats and oils, National Research Centre, Dokki, Cairo, Egypt
LiveDNA: 20.11675
Shaimaa Elsayed Mohammed
Department of Nutrition and Food Sciences, National Research Centre, Dokki, Cairo, Egypt
Mahmoud El-Sayed Ottai
Department of Genetics and Cytology, National Research Centre, Dokki, Cairo, Egypt