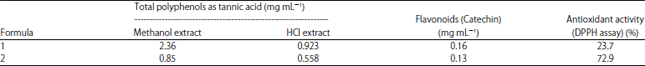
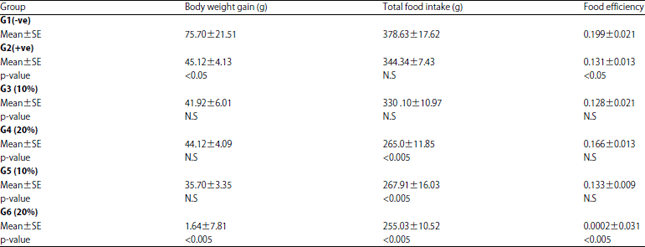
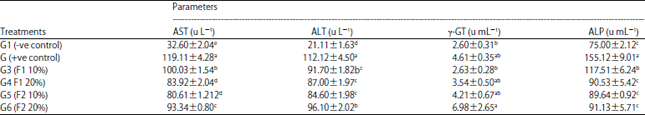
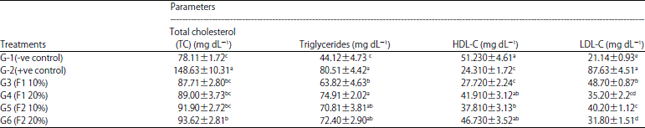
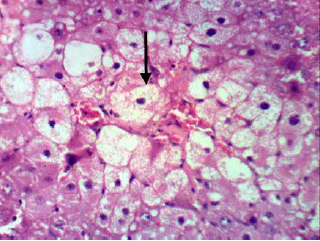
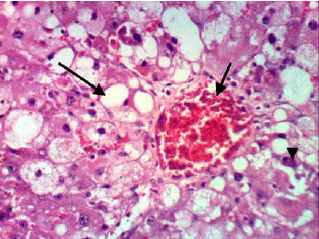
Research Article
Prevention from Complications Associating Liver Toxicity by Carbon Tetrachloride Using a Blend of Plants in the Form of Dietary Supplement
Department of Nutrition and Food Science, National Research Centre, Dokki, Giza, Egypt
LiveDNA: 20.16757
Amal S. Abdel-Azeem
Department of Nutrition and Food Science, National Research Centre, Dokki, Giza, Egypt
LiveDNA: 20.16759
Amany M. Hegazy
Department of Nutrition and Food Science, National Research Centre, Dokki, Giza, Egypt
I.H. Badawy
Department of Nutrition and Food Science, National Research Centre, Dokki, Giza, Egypt