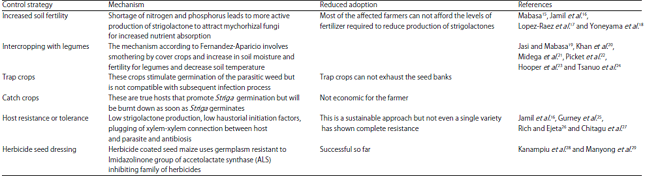
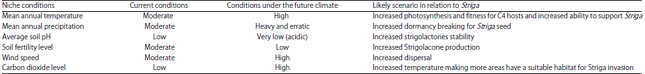
Perspective
Predictions of the Striga Scourge under New Climate in Southern Africa: A Perspective
Department of Agronomy, University of Fort Hare, P.O. Box X1314, 5700 Alice, South Africa
LiveDNA: 263.12613
Mutengwa Charles
Department of Agronomy, University of Fort Hare, P.O. Box X1314, 5700 Alice, South Africa
Mabasa Stanford
Department of Crop Science, University of Zimbabwe, P.O. Box MP167, Mount Pleasant, Harare, Zimbabwe
Mwenje Eddie
Department of Crop Science, Bindura University of Science Education, P.O. Box 1020, Bindura, Zimbabwe