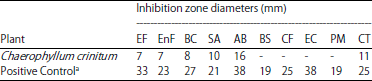
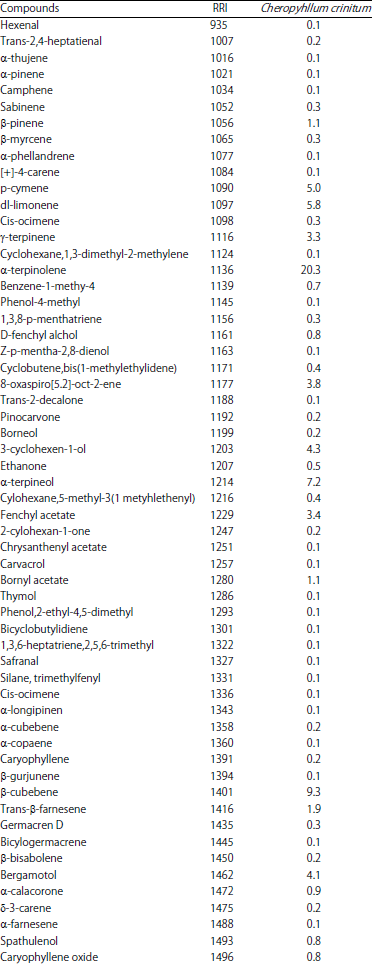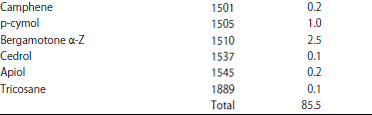Research Article
Evaluation of Essential Oil Composition, Antioxidant and Antimicrobial Properties of Chaerophyllum crinitum Boiss (Apiaceae) from Turkey: A Traditional Medicinal Herb
Department of Environmental Engineering, Faculty of Engineering and Architecture, Bitlis Eren University, Bitlis 13000, Turkey
Fatih Caglar Celikezen
Department of Chemistry, Faculty of Science and Letter, Bitlis Eren University, Bitlis 13000, Turkey