Research Article
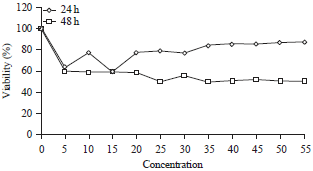
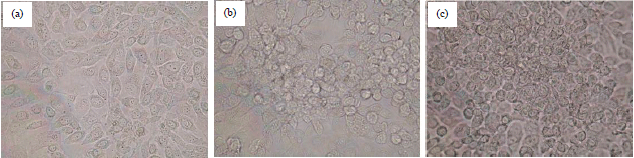
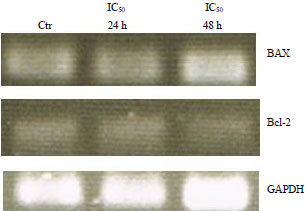
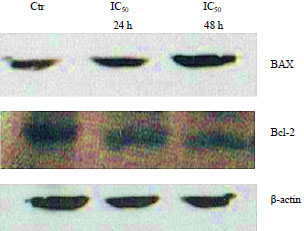
Antiproliferation of Fibrosarcoma Cell Line (L929) by Hua-Khao-Yen-Tai Thai Medicinal Plant (Dioscorea membranacea Pierre)
Department of Biology, Faculty of Science and Technology, Thepsatri Rajabhat University, Meuang, 15000 Lopburi, Thailand
Parisatcha Sangsuwan
Department of Biology, Faculty of Science and Technology, Thepsatri Rajabhat University, Meuang, 15000 Lopburi, Thailand