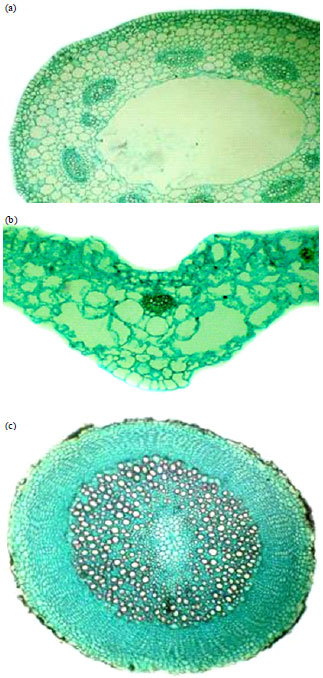
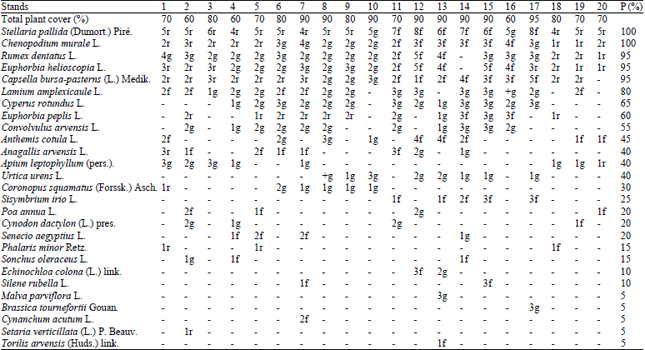
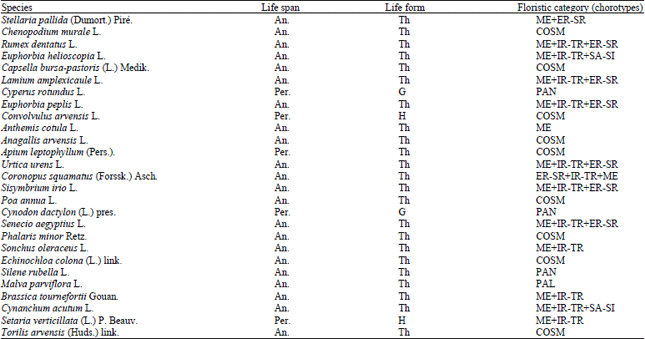
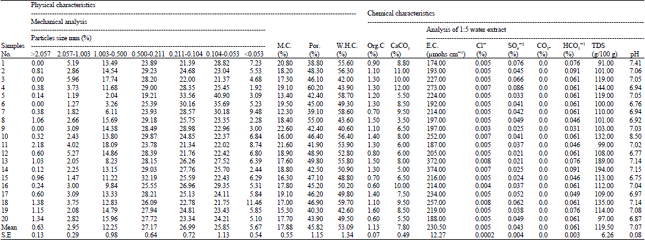
Research Article
Autecology and Bioactive Metabolites of Stellaria pallida Growing in North East Nile Delta, Egypt
Department of Botany, Faculty of Science, Mansoura University, Egypt
G.A. El Sherbeny
Department of Botany, Faculty of Science, Mansoura University, Egypt
H. M. Al-Jubawy
College of Education, Thi Qar University, Iraq