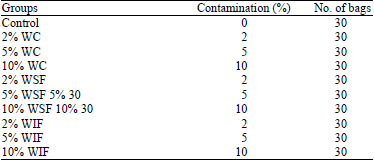
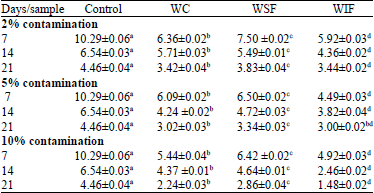
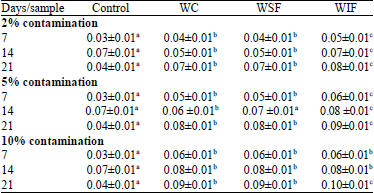
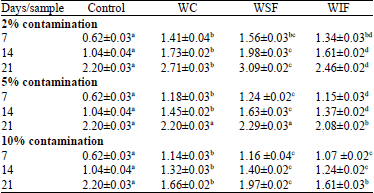
Research Article
Glucose-6-Phosphate Dehydrogenase and Carbohydrate in Bean (Vigna unguiculata) Exposed to Crude Oil
Department of Medical Biochemistry, School of Basic Medical Sciences, University of Benin, Nigeria
George E. Eriyamremu
Department of Biochemistry, Faculty of Life Sciences, University of Benin, Nigeria