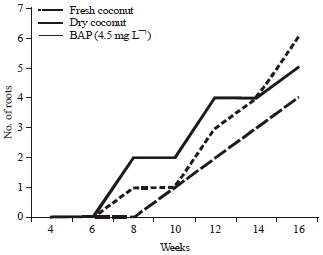
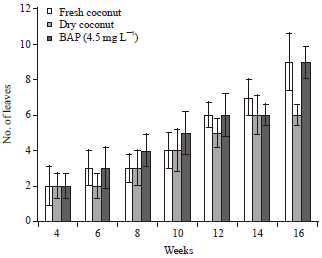
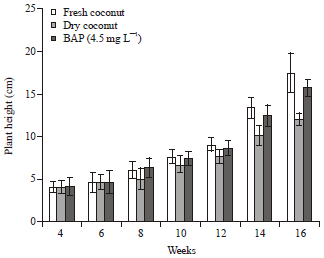
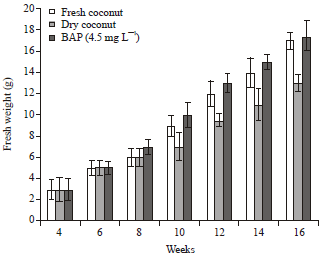
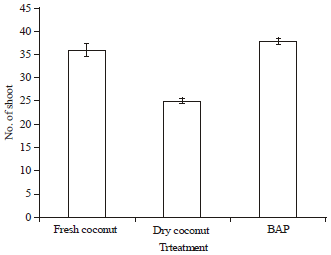
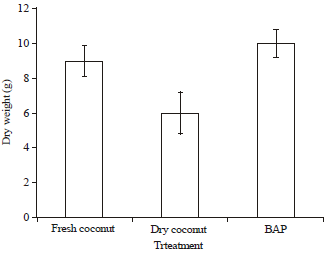
Research Article
Coconut Water from Fresh and Dry Fruits as an Alternative to BAP in the in vitro Culture of Dwarf Cavendish Banana
Department of Crop Science, School of Agriculture, University of Cape Coast, Ghana A R
P. Agu-Asare
Department of Crop Science, School of Agriculture, University of Cape Coast, Ghana A R