Research Article
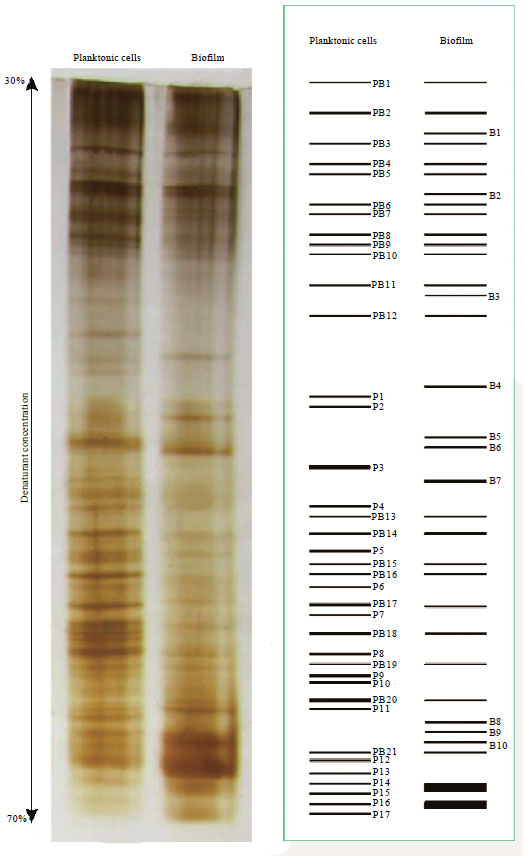
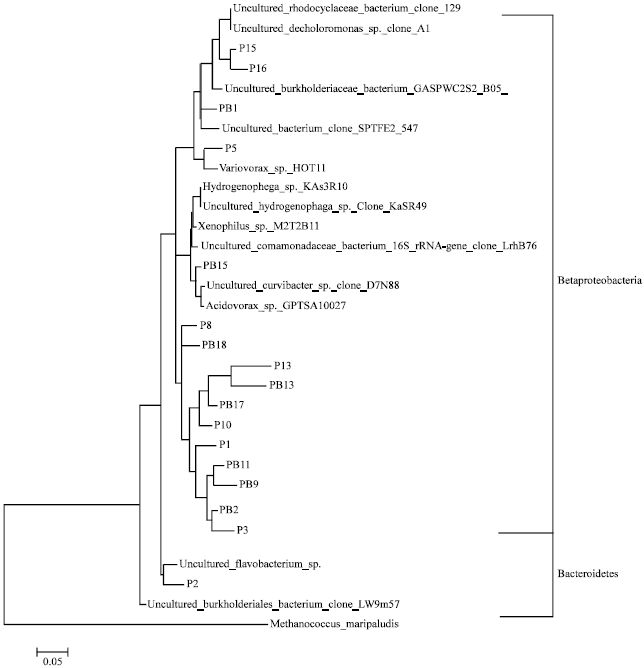
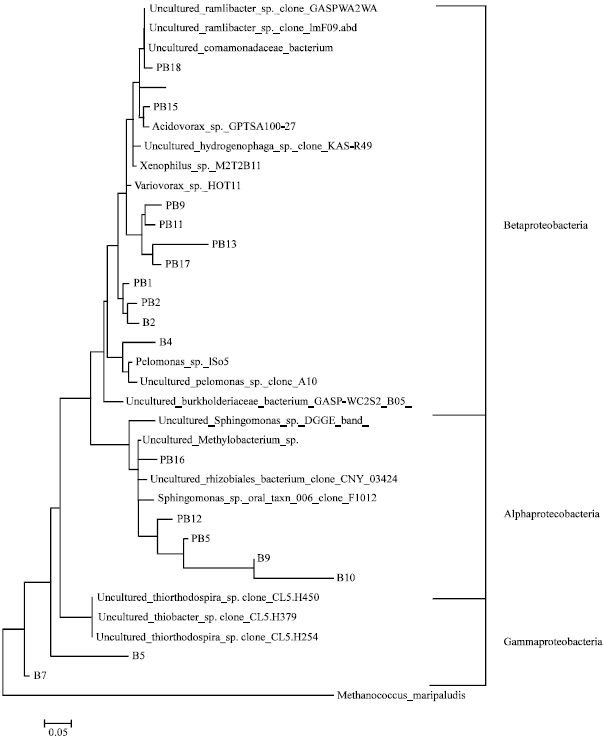
Bacterial Community Structures of Planktonic Cells and Biofilm at Saguling Hydro Power using Denaturing Gradient Gel Electrophoresis (DGGE)
School of Life Sciences and Technology, Institute of Technology Bandung, Jl. Ganesa 10 Bandung, Indonesia
Nur F. Afianti
School of Life Sciences and Technology, Institute of Technology Bandung, Jl. Ganesa 10 Bandung, Indonesia
Dea Indriani Astuti
School of Life Sciences and Technology, Institute of Technology Bandung, Jl. Ganesa 10 Bandung, Indonesia
Gede Suantika
School of Life Sciences and Technology, Institute of Technology Bandung, Jl. Ganesa 10 Bandung, Indonesia
Pingkan Aditiawati
School of Life Sciences and Technology, Institute of Technology Bandung, Jl. Ganesa 10 Bandung, Indonesia