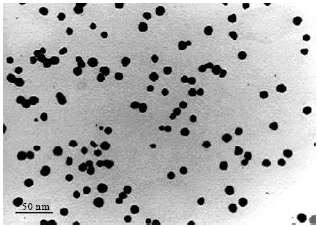
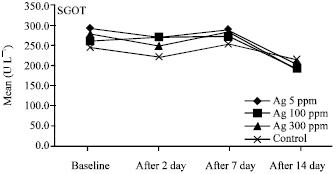
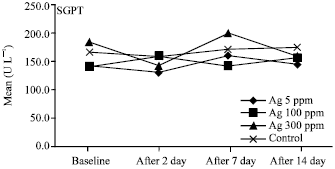
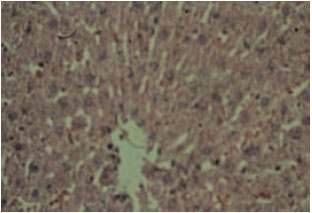
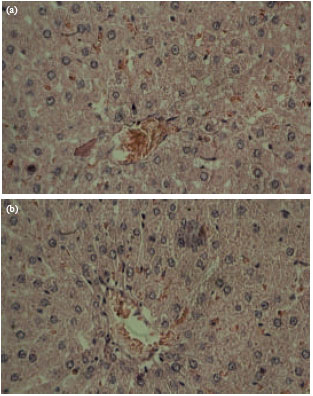
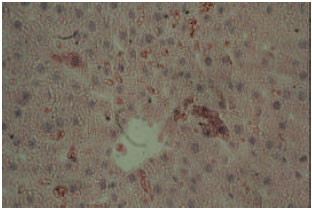
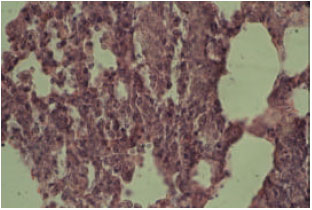
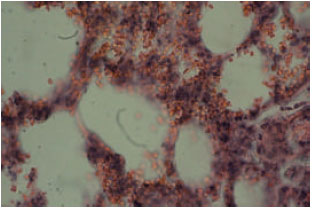
Research Article
Acute Effect of Nanosilver to Function and Tissue Liver of Rat after Intraperitioneal Injection
Department of Microbiology, Falavarjan Branch, Islamic Azad University, Isfahan, Iran
Mahbubeh Setorki
Department of Biology, Izeh Branch, Islamic Azad University, Izeh, Iran