Research Article
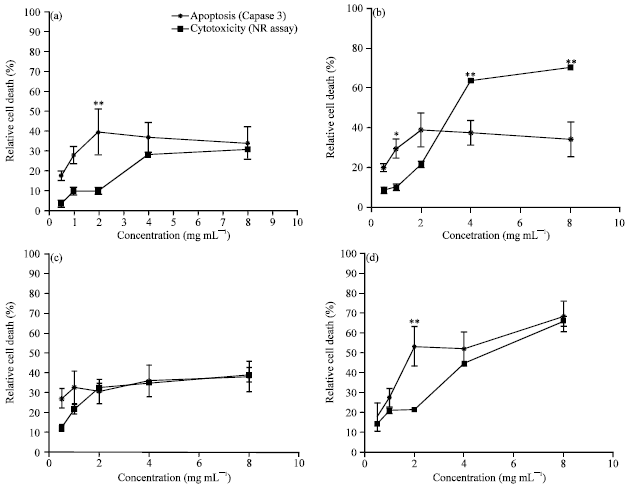
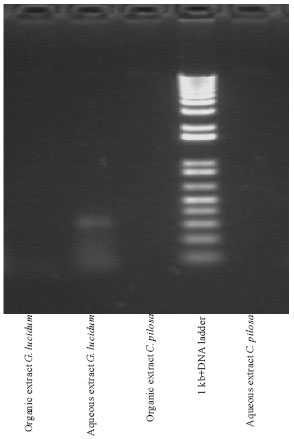
Cytotoxic and Apoptotic Potentials of Ganoderma lucidum and Curculigo pilosa on Human Cervical Adenocarcinoma Cell Line, HeLa
Department of Biochemistry, College of Medicine, University of Lagos, P.M.B. 12003, Lagos, Nigeria
Odesanmi O. Selina
Department of Biochemistry, College of Medicine, University of Lagos, P.M.B. 12003, Lagos, Nigeria
James Ayorinde Babatunde
Department of Biochemistry, College of Medicine, University of Lagos, P.M.B. 12003, Lagos, Nigeria
Tafida Mundi
Department of Biochemistry, College of Medicine, University of Lagos, P.M.B. 12003, Lagos, Nigeria
Olubunmi A. Magbagbeola
Department of Biochemistry, College of Medicine, University of Lagos, P.M.B. 12003, Lagos, Nigeria