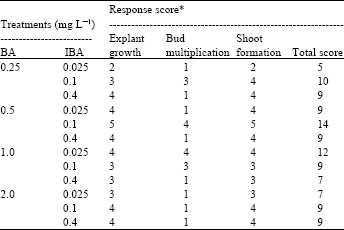
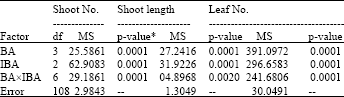
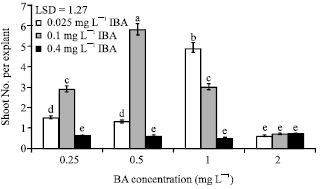
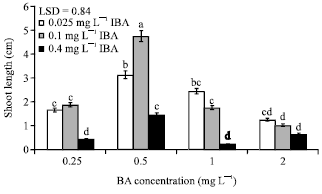
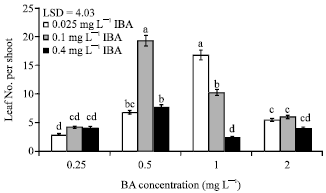
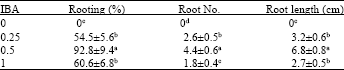
Research Article
Conservation of Endangered Hassawi Peach (Prunus persica L.) Through Micropropagation
Department of Agricultural Biotechnology, College of Agricultural and Food Sciences, King Faisal University, P.O. Box 420, Al-Hassa 31982, Saudi Arabia
J.M. Al-Khayri
Department of Agricultural Biotechnology, College of Agricultural and Food Sciences, King Faisal University, P.O. Box 420, Al-Hassa 31982, Saudi Arabia