Mini Review
Theobromine Toxicity and Remediation of Cocoa By-products: An Overview
Department of Biochemistry, Cell and Molecular Biology, University of Ghana, P.O. Box LG 54, Legon, Ghana
World production of cocoa (Theobroma cacao) beans, primarily for the manufacture of chocolate and cocoa powder, currently stands at 4.05 million metric tonnes (International Cocoa Organization, 2012). Countries in West and Central Africa account for 71.4% of total production, and generate an estimated 6.7 million metric tonnes of cocoa pod husk, a by-product of cocoa cultivation, as well as cocoa bean shell and cocoa bean cake, which are waste products of chocolate production. Given the critical shortfall in livestock production in most cocoa-producing countries in Africa, attributable largely to the prohibitive cost of animal feed, the utilization of the vast quantities of discarded cocoa waste products as affordable accessory animal feed would be of tremendous benefit to millions (Owusu-Domfeh, 1972; Alemawor et al., 2009; Ogunlade et al., 2010; Hamzat and Adeola, 2011).
Unfortunately, however, the results of feeding trials have been less than encouraging. Many have provided evidence of toxicity at high dietary concentrations of cocoa waste materials, necessitating research on detoxification measures. 3,7-Dimethyl-3,7-dihydro-1H-purine-2,6-dione, commonly known as theobromine (Fig. 1), is believed to be responsible for most of the toxic effects of cocoa by-products.
The purpose of this mini-review is to examine the evidence implicating theobromine as the toxic factor in cocoa by-products, and to highlight progress made in the development of procedures for its elimination from cocoa by-products.
 | |
| Fig. 1: | Structure of Theobromine (source: http://upload.wikimedia.org/wikipedia/commons/thumb/b/b1/Theobromin_-_Theobromine.svg/194px-Theobromin_-_Theobromine.svg.png) |
FEED POTENTIAL OF COCOA BY-PRODUCTS
A comparative analysis of cocoa pod husk and natural fodder shows the proximate composition profiles of the two compare favorably, and suggests that the husk has considerable potential as non-conventional complementary ruminant feedstuff (Table 1). In addition, owing to the presence of pectin and free sugars, cocoa pod husk could supply a substantial amount of the energy requirements of non-ruminants, although its high fibre content appears to depress feed utilization by non-ruminants (Longe and Ogedegbe, 1989; Alemawor et al., 2009). It is worth noting that the nutrient profile of cocoa pod husk is also very similar to that of soybean hulls, which are routinely included in animal diets in North America. As shown in Table 1, both cocoa bean shell and bean cake are rich in protein (14-29%), a vital component of animal feed. Given that poultry, pigs and ruminants require 14-18, 13-26 and 10% protein respectively, virtually all of their protein maintenance requirements could be derived from these cocoa by-products.
| Table 1: | Comparison of selected constituents of cocoa by-products |
 | |
| Owusu-Domfeh (1972), Odunsi and Longe (1998), Adamafio et al. (2004), Laconi (2009) and Feedipedia (2012) | |
EFFECTS OF COCOA BY-PRODUCTS ON LIVESTOCK
Attempts to utilize cocoa waste materials as feed resources have shown that, often, when dietary concentrations exceed 10-15%, growth and reproductive indices are negatively affected (Ettlin et al., 1986; Odunsi and Longe, 1995; Yang et al., 1997; Adeyina et al., 2010). The consumption of organic mulch, composed of cocoa by-products, is reported to cause vomiting, central nervous system depression, restlessness, diarrhoea, muscle tremor, ataxia, haematuria, tachycardia and seizures in animals (Tarka Jr. et al., 1981; Gans, 1984; Day and Dilworth, 1984; Drolet et al., 1984). Cases of mortality have also been documented (Alexander et al., 2008). However, susceptibility to the detrimental effects induced by cocoa by-products appears to be species-dependent and age-dependent.
Ruminants: An early study showed that the inclusion of 9% cocoa shell in the diets of lambs stimulated feed intake and growth. However, higher inclusion rates caused a reduction in feed intake and weight gain (Tarka Jr et al., 1981). Others observed a reduction in body weight when cocoa shell was included in the daily ration of sheep and goats. This phenomenon was reversed when cocoa materials were excluded from the diet (Alexander et al., 2008). In evaluating the consequences of cocoa waste consumption by livestock, scant attention has been paid to ruminants in recent times.
Pigs: A number of studies have shown that the inclusion of up to 30% cocoa pod husk in the diet of growing-finishing pigs does not cause any detrimental effects (Oddoye et al., 2010). Although, earlier studies had found that feeding of cocoa pod husk led to spontaneous abortions in pregnant pigs, Rhule et al. (2005) reported that 20% cocoa pod husk diets did not adversely affect the reproductive performance of breeding sows. However, the researchers observed that the inclusion of 20% cocoa pod husk, but not 10%, reduced the neonatal weight of piglets.
Poultry: A number of researchers have reported poor performance following the ingestion of cocoa materials by chickens. Egg production was adversely affected by the consumption of cocoa bean shell in a study conducted by Olubamiwa et al. (2006). Teguia et al. (2004) also observed detrimental effects on growth when the level of cocoa pod husk incorporated in the diet of broiler chickens exceeded 10%. According to Odunsi et al. (1999), broiler chickens fed 15% untreated cocoa bean meal exhibited negative effects including an increase in creatinine levels and a reduction in feed intake, weight gain and haemoglobin level. These adverse effects were absent in chickens fed a similar amount of cocoa bean meal which had been treated with alkali or hot water to reduce theobromine content. However, detrimental effects were observed at an inclusion rate 30% of alkali-treated or hot water-treated cocoa bean meal. These observations are in conformity with the findings of earlier studies, in which Odunsi and Longe (1995, 1998) observed depressed weight gain and feed intake as well as increased mortality when chickens were fed 10-30% cocoa bean meal. Similar observations have been made following the inclusion of cocoa shell meal in the diets of broiler chickens (Day and Dilworth, 1984).
THEOBROMINE TOXICITY
Extensive studies conducted to investigate the underlying causes of these phenomena have identified theobromine (3,7-dimethylxanthine) as the principal causative factor (Gans, 1984; Tarka et al., 1986; Wang et al., 1992; Alexander et al., 2008). Much of the evidence implicating theobromine as a toxic factor in animals has come from studies involving the dietary administration of pure theobromine to laboratory animals and livestock. These studies have demonstrated that the methylxanthine induces a wide range of aberrations, which can be divided into two broad categories: reproductive and developmental toxicity.
Reproductive toxicity: In studies conducted under the auspices of the National Institute of Environmental Health Sciences of the United States, theobromine administered in feed severely impaired the reproductive capacity of female Swiss CD-1 mice, as evidenced by an increase in the number of lifeless pups per litter (NTP, 1984). In males, the incidence of abnormal sperm was significantly increased while testicular weight was reduced. Theobromine-induced alterations in Sertoli cells and in testicular morphology have also been documented (Wang et al., 1992; Ying and Waller, 1994).
Developmental toxicity: Other detrimental effects of theobromine include a reduction in thymus weight, a slow heart rate, excitability, digestive complications and impaired growth (Howell et al., 1997; Alexander et al., 2008). Chronic theobromine poisoning can result in epileptic-like seizures or death (Mitchell et al., 2011). According to Soffietti et al. (1989), the inclusion of 1-1.5% theobromine in the diets of immature rabbits for 120 days caused severe and rapid mortality.
The elimination of theobromine from cocoa by-products is, therefore, critical to their utilization as animal feed. Theobromine is ubiquitous in the cocoa plant (Zheng et al., 2004), but its concentration varies from one part of the plant to another, as shown in Table 2.
| Table 2: | Theobromine content of cocoa products |
 | |
| Owusu-Domfeh (1972), Sotelo and Alvarez (1991) Odunsi and Longe (1998), Pura (2001) and Laconi (2009) | |
Geographical and varietal differences in theobromine content have been documented, with cocoa from Africa recording the highest theobromine content.
Clearly, the development of appropriate detheobromination methodologies would greatly enhance utilization of the vast quantities of cocoa pod husk and other cocoa by-products generated annually in cocoa-producing countries (Adamafio et al., 2011; Smith, 2011). This is critical to the realization of the enormous economic and environmental benefits to be derived from their utilization as feed resources.
SPECIES-DEPENDENT TOXICITY
Interestingly, there are no reported cases of theobromine toxicity in humans, despite the consumption of chocolate confectioneries for centuries. Indeed, theobromine is currently regarded as a nutraceutical for humans. Even among animals, some species such as dogs and horses appear to be far more sensitive to the adverse effects of the methylxanthine than others are. The reasons for this are not entirely clear, but may lie in inter-species differences in the rate of theobromine catabolism.
THEOBROMINE CATABOLISM
Multiple routes exist by which the deconstruction of theobromine may occur in biological organisms (Dash and Gummadi, 2006; Mazzafera et al., 1999). As shown in Fig. 2, in some instances, catabolism involves sequential N-demethylation to either 7-methylxanthine or 3-methylxanthine then to xanthine and subsequent oxidation to uric acid in the liver (Blecher and Lingens, 1977; Dash and Gummadi, 2006).
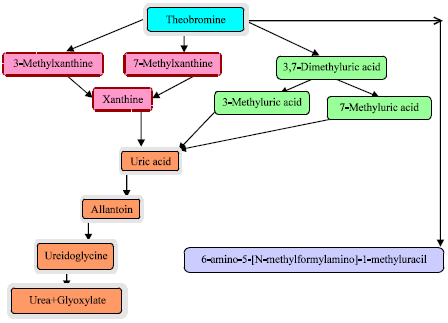 | |
| Fig. 2: | Theobromine degradation pathways |
Alternatively, theobromine can be oxidized directly to 3,7-dimethyluric acid in a reaction catalyzed by theobromine oxidase, theobromine dehydrogenase or a broad substrate specificity xanthine oxidase (Yu et al., 2009), and subsequently converted to uric acid. A number of cytochrome P450 enzymes (e.g., CYP1A2 and CYP2E1), N-acetyltransferase and xanthine oxidase participate in the degradation process (Gates and Miners, 1999). The profile of final degradation products of theobromine metabolism is species-dependent. Although the catabolic pathway in humans is similar to that described in many animal species, huge differences in rates have been documented. For instance, plasma half-lives in livestock range from 15.5-21 h compared with 7-12 h for humans (Tarka Jr.et al., 1981; Shively et al., 1985, Lelo et al., 1986). The relatively low rate of degradation may account for the adverse effects of theobromine on animals.
DETHEOBROMINATION METHODS
Attempts have been made to detoxify cocoa by-products using physicochemical and biological methods. A measure of success has been achieved using both types of methods.
Physicochemical treatment: Olubamiwa et al. (2006) investigated the effect of boiling on cocoa bean shell composition, and observed time-dependent detheobromination. However, a feeding trial showed that although the performance of chickens improved when cocoa bean shell was boiled for 15 minutes, prolonged boiling produced poor results. This is hardly surprising since prolonged boiling would render the shells less nutritive by extracting soluble nutrients such as sugars and proteins, in addition to the theobromine. Odunsi et al. (1999) also found that treatment of cocoa bean cake with either hot water (90oC) or alkali (cocoa-pod ash solution) reduced theobromine content by 56% and 72% respectively. Both alkali and hot water treatment also caused decreases in lipid, mineral element and metabolisable energy values. In addition, alkali treatment resulted in a significant reduction in protein content. Nutritional evaluation of both hot water-treated and alkali-treated cocoa bean cake showed improved food utilisation and nitrogen retention by broiler chickens at low dietary concentrations. However, growth performance at high dietary concentrations of the treated bean cake was considered unsatisfactory (Odunsi and Longe, 1998).Studies on cocoa bean shells have produced similar findings (Adeyina et al., 2008, 2010). Studies have shown that as much as 60.7% of soluble sugars is lost when cocoa pod husk is treated with dilute alkali (unpublished observations). In all likelihood, the loss of vital soluble nutrients, such as proteins, sugars and micronutrients, during treatment with hot water or alkali, contributed to the poor performance of the poultry at high dietary concentrations.
Biological treatment: Microbial treatment is a promising alternative to physicochemical procedures, and circumvents the loss of soluble nutrients. The isolation of atoxigenic microorganisms that express theobromine-degrading enzymes and have the capacity to colonize cocoa by-products is critical to the successful development of biological detheobromination methods.
Filamentous fungi: Fungal treatment of cocoa bean shell has been investigated by Muhammed et al. (2000), who reported that treatment with Pleurotus pulmonarium for four weeks significantly reduced theobromine content of cocoa bean shell by 24.5% and increased protein content. Albino rats were maintained on diets containing no cocoa bean shell or diets in which 5% of maize had been replaced by untreated or fungi-treated cocoa bean shell. Rats on the P. pulmonarium-treated bean shell recorded a higher growth rate; 12.2% compared with 9.1% for untreated bean shell. While these findings are encouraging, it would be useful to investigate growth performance at inclusion rates that exceed 10% in order to determine whether the bioprocess eliminates theobromine toxicity. In another study, microorganisms associated with cocoa pods were screened for the capacity to degrade theobromine (Adamafio et al., 2011). A. niger emerged the dominant theobromine-metabolizing organism isolated from the pods. After sub-culturing to obtain single colonies, cocoa pod husk samples were inoculated with A. niger isolates. Solid-state fermentation for 7 days resulted in a 71.8% reduction in theobromine content (Adamafio et al., 2011). These findings on A. niger remediation, although promising, would only prove useful if the possibility of ochratoxin A contamination could be avoided or minimized (Varga et al., 1996; Klich et al., 2009). The nephrotoxic and teratogenic effects of ochratoxin A on livestock, as well as its persistence in the food chain, would be matters of particular concern if A. niger were to be used for the bioremediation of theobromine-containing materials.
Several strategies exist for dealing with possible ochratoxin A contamination during A. niger bioremediation. A large number of ochratoxin A-degrading fungal and bacterial strains including Rhizopus stolonifer, R. oligosporus, Lactobaccillus acidophilus and Acinobacter calcoaceticus have been studied extensively. Their capacity to degrade the toxin in liquid media is well documented (Varga et al., 2005; Abunrosa et al., 2006). Co-culturing and sequential-culturing of cocoa waste materials with A. niger and ochratoxin A-degrading isolates might render detoxified cocoa by-products acceptable for use at high dietary concentrations. An alternative strategy would be to explore the possibility of degrading theobromine through the direct application of cell-free A. niger extracts to cocoa by-products. Data would need to be generated on the stability, activity and effective concentrations of theobromine demethylases in the cell-free extracts. This approach might be advantageous, but is yet to be explored.
Yeasts and bacteria: Adamafio et al. (2012) have isolated a novel theobromine-degrading Candida krusei strain. Although C. krusei-induced detheobromination appears to be slow in comparison with the rates exhibited by A. niger, the use of C. krusei would avoid possible ochratoxin A contamination. Another potential biological tool is Pseudomonas putida CBB5, which has been shown to degrade all known methylxanthines in liquid medium (Yu et al., 2009). Its suitability as a biodetheobromination tool for cocoa by-products is yet to be investigated.
Challenges and future directions: Much remains to be done in the development of appropriate detheobromination technologies for cocoa by products. Comparative studies on isolated strains of theobromine-degrading organisms such as C. krusei and P. putida CBB5 will be necessary to identify the most efficacious biodetheobromination tools. The optimal process parameters for solid-state fermentation will need to be described for each potential biological tool. Also, it would be worthwhile to continue to bioprospect for novel atoxigenic theobromine-degrading microorganisms.
In conclusion, while progress has been made in the development of detheobromination procedures, existing physicochemical methods need to be modified to minimize the loss of soluble nutrients, in order to ensure satisfactory performance by non-ruminants at high dietary concentrations of treated cocoa by-products. Although biodetheobromination methods appear to be attractive alternatives to physicochemical methods, extensive studies need to be conducted to ensure that the mycotoxin levels of final products do not exceed the permissible limits for livestock.
omosogbe folagbade Reply
This is an a way forward for many researcher willing to cut cost in production.