Research Article
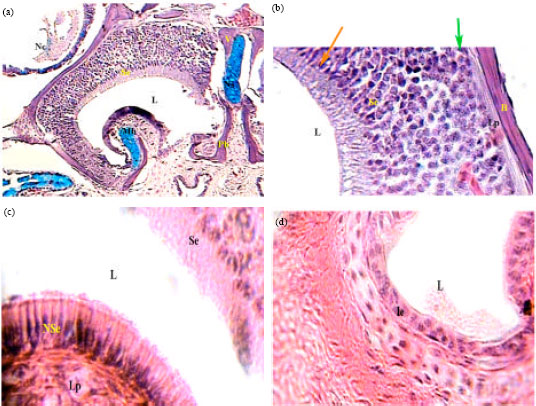
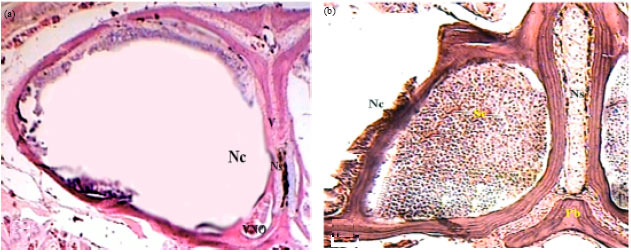
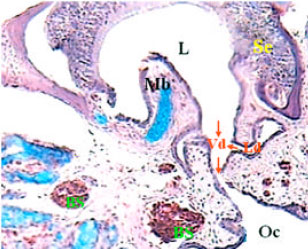
Histological Studies on the Vomeronasal Organ of the Worm-like Snake, Typhlops vermicularis
Department of Biology, Faculty of Science, Lorestan University, Khorramabad, Iran
Mohsen Abbasi
Department of Basic Science, Faculty of Veterinary Medicine, Lorestan University, Khorramabad, Iran
Parisa Yusefi
Department of Biology, Faculty of Science, Lorestan University, Khorramabad, Iran