Research Article
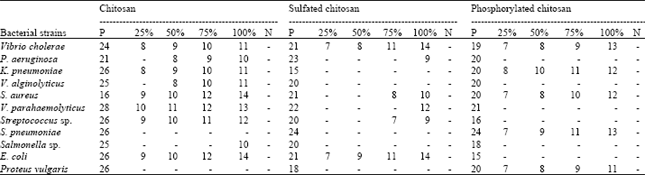
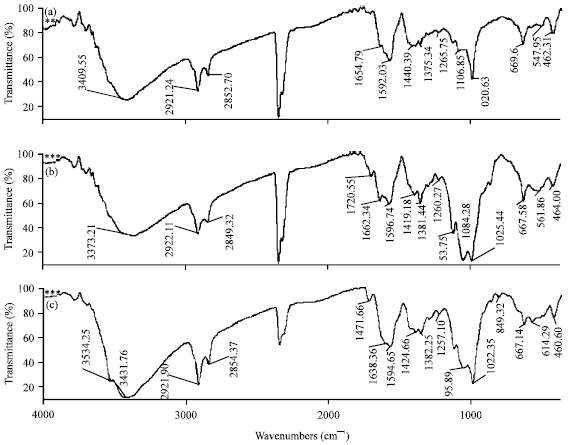
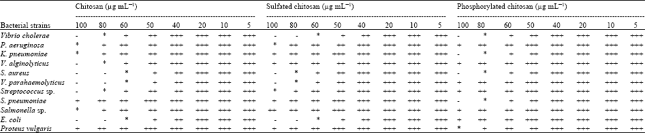
Preparation of Chitosan Derivatives from Gladius of Squid Sepioteuthis lessoniana (Lesson, 1830) and Antimicrobial Potential against Human Pathogens
CAS in Marine Biology, Faculty of Marine Sciences, Annamalai University, Parangipettai, 608502, India
Pasiyappazham Ramasamy
CAS in Marine Biology, Faculty of Marine Sciences, Annamalai University, Parangipettai, 608502, India
Alagiri Srinivasan
Department of Biophysics, All India Institute of Medical Sciences, New Dehli, India
Annaian Shanmugam
CAS in Marine Biology, Faculty of Marine Sciences, Annamalai University, Parangipettai, 608502, India
Vairamani Shanmugam
CAS in Marine Biology, Faculty of Marine Sciences, Annamalai University, Parangipettai, 608502, India