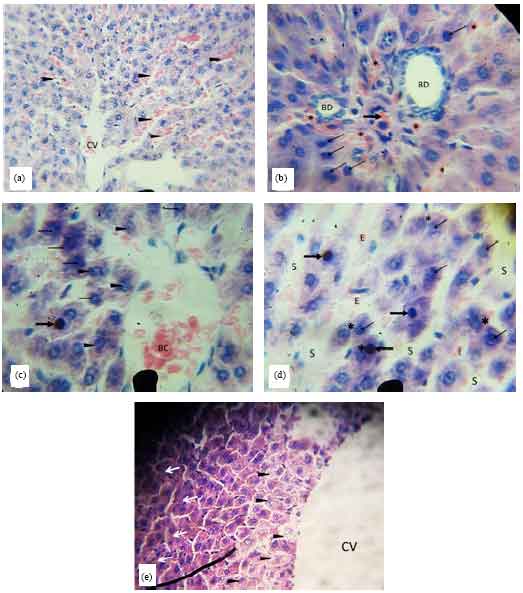
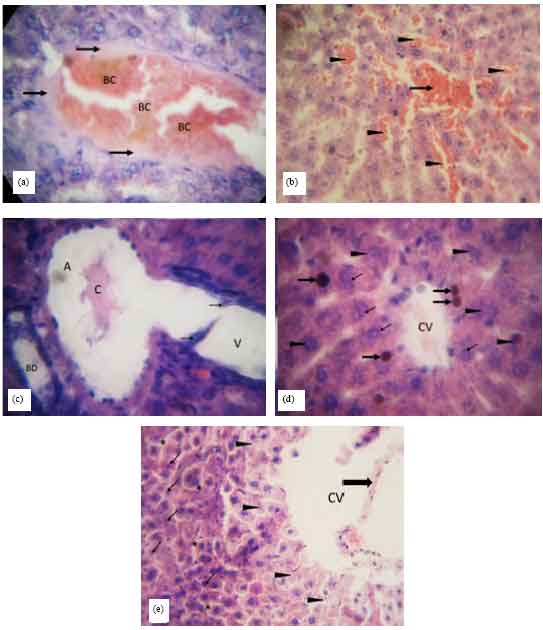
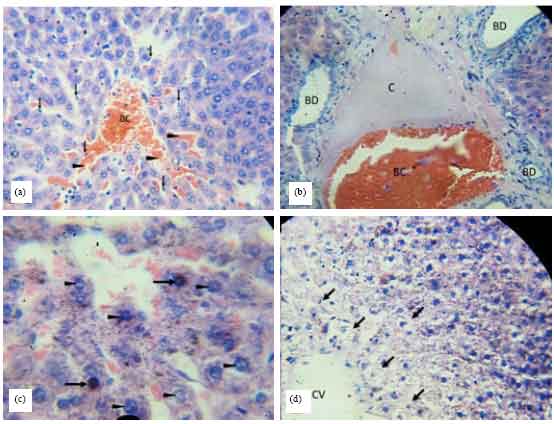
Research Article
Enzyme Activity and Histopathology of Rat Liver Treated with Crude Methanolic Extract of Pepperomia pellucida (L.) HBK
Department of Biology, College of Arts and Sciences, University of the Philippines Manila, Padre Faura St. Taft Avenue, Ermita, Manila, Philippines
Elisa L. Co
Department of Biology, College of Arts and Sciences, University of the Philippines Manila, Padre Faura St. Taft Avenue, Ermita, Manila, Philippines
Sally Andrea D. Gaspi
Department of Biology, College of Arts and Sciences, University of the Philippines Manila, Padre Faura St. Taft Avenue, Ermita, Manila, Philippines
Jessica Leika R. Matibag
Department of Biology, College of Arts and Sciences, University of the Philippines Manila, Padre Faura St. Taft Avenue, Ermita, Manila, Philippines
Glenn L. Sia Su
Department of Biology, College of Arts and Sciences, University of the Philippines Manila, Padre Faura St. Taft Avenue, Ermita, Manila, Philippines