Research Article
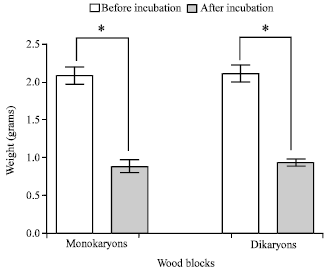
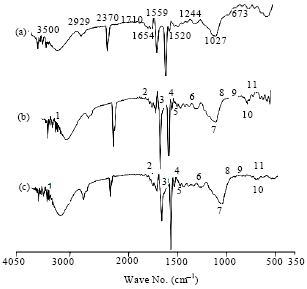
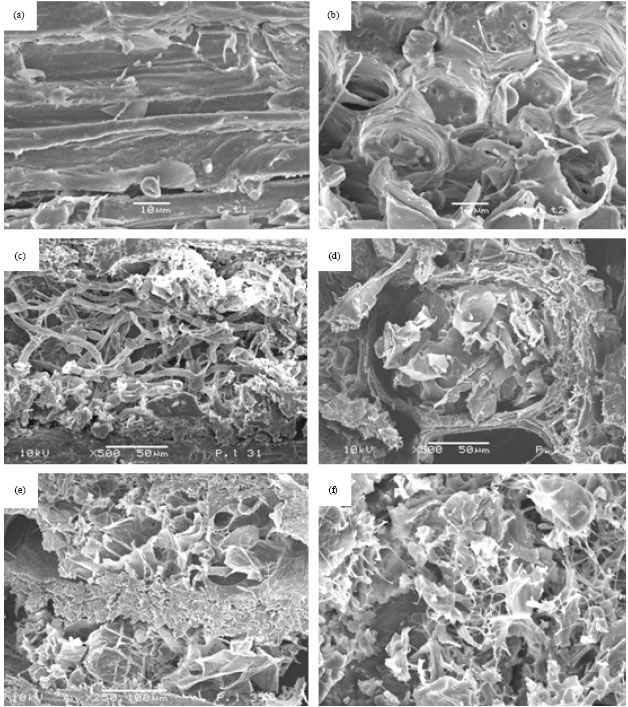
Comparison of Differences Between the Wood Degradation by Monokaryons (n) and Dikaryons (2n) of White Rot Fungus (Cambodian Phellinus linteus)
Department of Biology, Faculty of Science, Mahasarakham University, Khamriang Sub-District, Kuntarawichai District, Mahasarakham Province, 44150, Thailand
Wasana Chaiseana
Faculty of Science and Agricultural Technology, Rajamangala University of Technology Lanna, Phisanuklok Province, Thailand
Panida Loutchanwoot
Department of Biology, Faculty of Science, Mahasarakham University, Khamriang Sub-District, Kuntarawichai District, Mahasarakham Province, 44150, Thailand
Piyarat Dornbundit
Department of Chemistry, Faculty of Science, Srinakarintarawirot University, Sukhumvit 23, Bangkok, Thailand