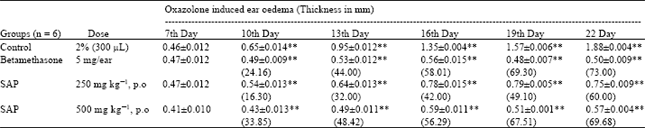
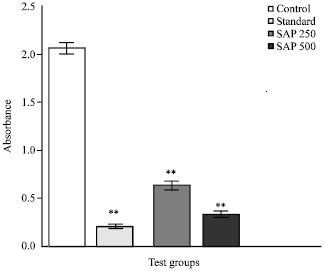
Research Article
Effect Of Saponins From Of Sesbania sesban L.(Merr) On Acute And Chronic Inflammation in Experimental Induced Animals
R.C. Patel Institute of Pharmaceutical Education and Research, Shirpur, Dhule (M.S) 425405, India
Payal R. Dande
SVKM, School of Pharmacy, Mumbai, Shirpur campus (M.S) 425405, India
Rakesh E. Mutha
H.R. Patel Institute of Pharmaceutical Education and Research, Shirpur, Dhule (M.S) 425405, India
Sanjay J. Surana
R.C. Patel Institute of Pharmaceutical Education and Research, Shirpur, Dhule (M.S) 425405, India