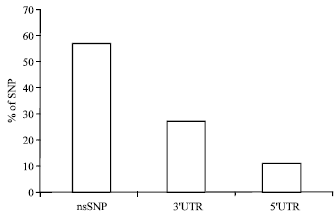
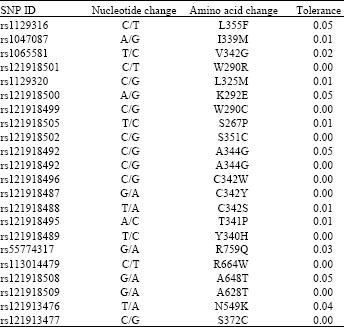
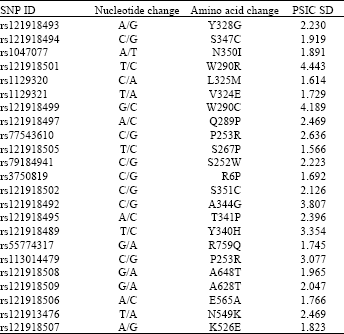
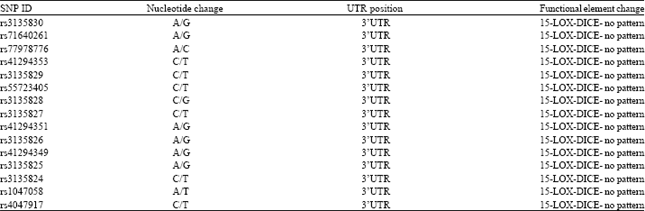
Research Article
In silico Analysis of Deleterious SNPs of the FGFR2 Gene
Department of Bioinformatics, Alagappa University, Karaikudi-630 003, Tamil Nadu, India
R. Vanajothi
Department of Bioinformatics, Alagappa University, Karaikudi-630 003, Tamil Nadu, India
A. Sudha
Department of Bioinformatics, Alagappa University, Karaikudi-630 003, Tamil Nadu, India
P. Rameshthangam
Department of Biotechnology, Alagappa University, Karaikudi-630 003, Tamil Nadu, India
P. Srinivasan
Department of Bioinformatics, Alagappa University, Karaikudi-630 003, Tamil Nadu, India