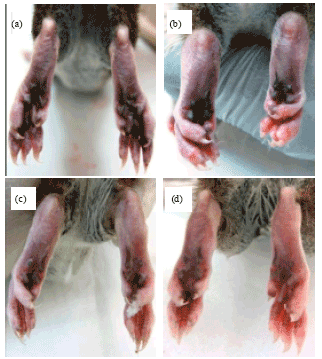
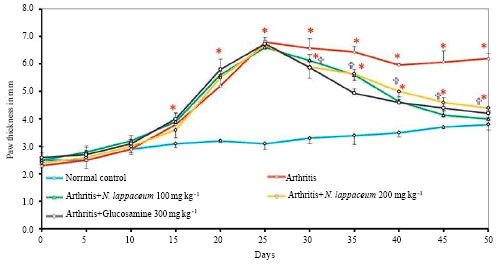
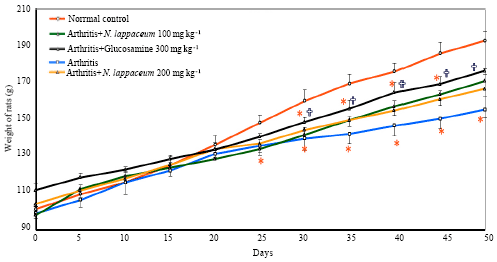
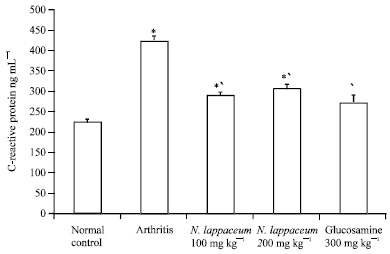
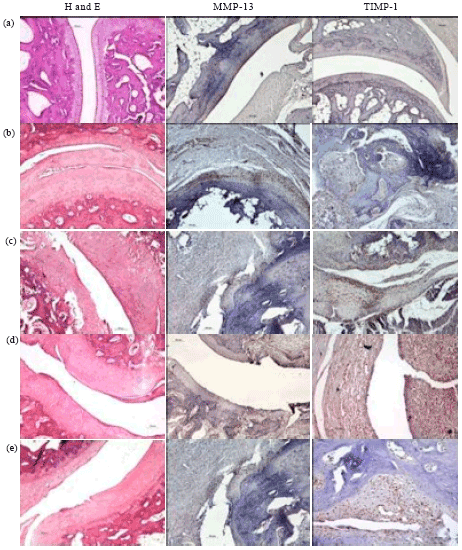
Research Article
Protective Effects of Nephelium lappaceum Rind Extract against Collagen-induced Arthritis in Dark Agouti Rats
International Medical University, Kuala Lumpur, Malaysia
Srikumar Chakravarthi
International Medical University, Kuala Lumpur, Malaysia
Gan Seng Chiew
Department of Pre-clinical Sciences, Faculty of Medicine and Health Sciences, UTAR, Malaysia
Thavamanithevi Subramaniam
SIRIM Bhd, Shah Alam, Malaysia
Umadevi Palanisamy
School of Medicine and Health Sciences, Monash University Sunway Campus, Malaysia
Ammu Radhakrishnan
International Medical University, Kuala Lumpur, Malaysia
Nagaraja Haleagrahara
Discipline of Physiology and Pharmacology, School of Veterinary and Biomedical Sciences, James Cook University, Australia