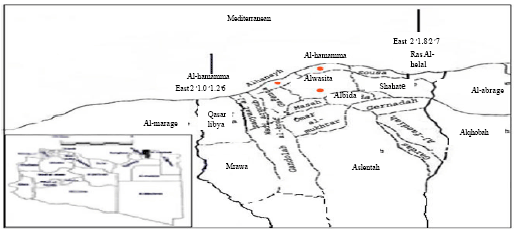
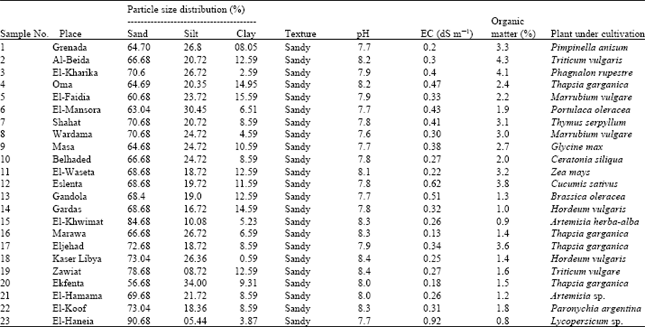
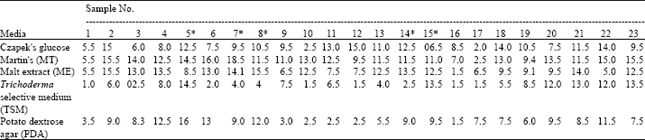
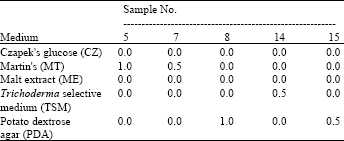
Research Article
Occurrence and Microbiological Characteristics of Trichoderma in Al-Jabal Al-Akhdar Region, Libya
Department of Microbiology, Faculty of Education, Omar Al-Mukhtar University, P.O. Box 919, Al-Bayda, Libya
Suliman S. Abdelrawaf
Department of Microbiology, Faculty of Education, Omar Al-Mukhtar University, P.O. Box 919, Al-Bayda, Libya
Khawila Saeed Omar
Department of Biology, Faculty of Education, Omar Al-Mukhtar University, P.O. Box 919, Al-Bayda, Libya
H.M.A. El-Komy
Department of Botany, Faculty of Education, Omar Al-Mukhtar University, P.O. Box 919, Al-Bayda, Libya
Muhammed Sarwar
Asian Network for Scientific Information, 308-Lasani Town, Sargodha Road, Faisalabad, Pakistan