Research Article
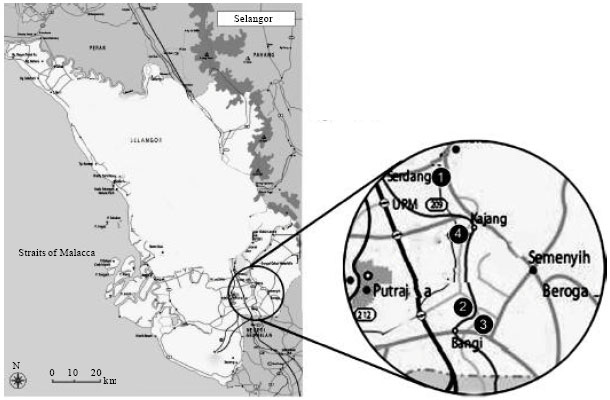
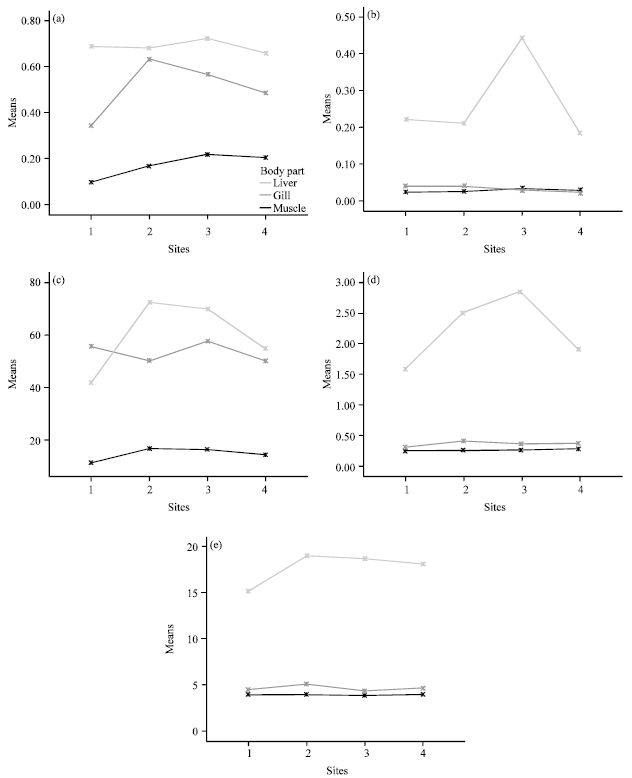
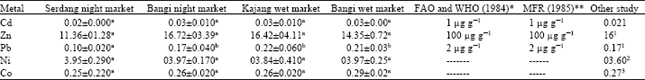
Analysis of Heavy Metal Concentrations in Tilapia Fish (Oreochromis niloticus) From Four Selected Markets in Selangor, Peninsular Malaysia
School of Environmental and Natural Resource Sciences, Faculty of Science and Technology, University Kebangsaan Malaysia, Bangi, 43600, Selangor
M. Shuhaimi-Othman
School of Environmental and Natural Resource Sciences, Faculty of Science and Technology, University Kebangsaan Malaysia, Bangi, 43600, Selangor
A.K. Ahmad
School of Environmental and Natural Resource Sciences, Faculty of Science and Technology, University Kebangsaan Malaysia, Bangi, 43600, Selangor












samadhi Wathsala Reply
I want read this article to improve my knowledge.