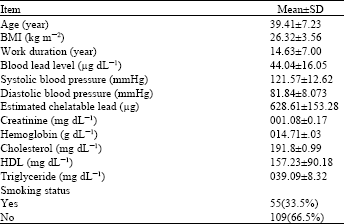
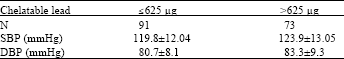
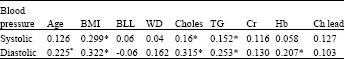
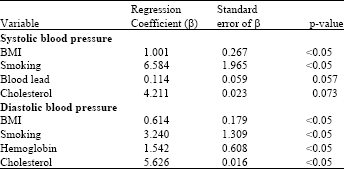
Research Article
The Relationship Between Blood Pressure and Lead Exposure in Battery Recycling Workers
Centre for Research on Occupational Diseases, Department of Occupational Medicine, Tehran University of Medical Sciences, Tehran, Iran
Leila S. Bahaedini
Centre for Research on Occupational Diseases, Department of Occupational Medicine, Tehran University of Medical Sciences, Tehran, Iran
Ramin Mehrdad
Centre for Research on Occupational Diseases, Department of Occupational Medicine, Tehran University of Medical Sciences, Tehran, Iran
Ali Pasha Meysami
Department of Community Medicine, Tehran University of Medical Sciences, Tehran, Iran
Farzaneh Chavoshi
Health Research Center, Baqiyatallah University of Medical Sciences, Tehran, Iran
Shahin Soltani
Centre for Research on Occupational Diseases, Department of Occupational Medicine, Tehran University of Medical Sciences, Tehran, Iran