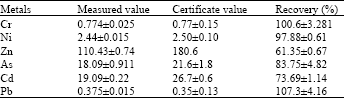
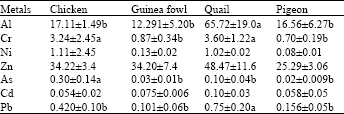
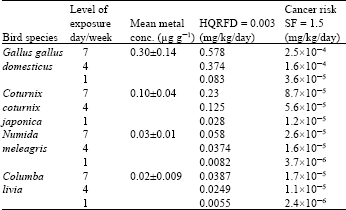
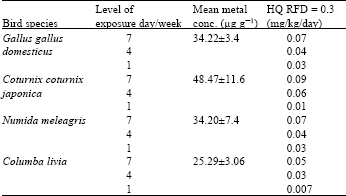
Research Article
Metals Concentrations in Eggs of Domestic Avian and Estimation of Health Risk from Eggs Consumption
School of Environmental and Natural Resource Sciences, Faculty of Sciences and Technology, National University of Malaysia, 43600, Bangi, Selangor, Malaysia
M. Shuhaimi- Othman
School of Environmental and Natural Resource Sciences, Faculty of Sciences and Technology, National University of Malaysia, 43600, Bangi, Selangor, Malaysia