Research Article
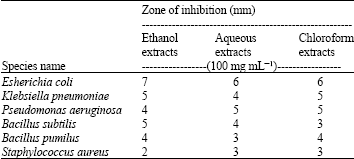
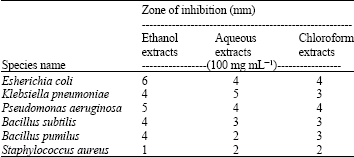
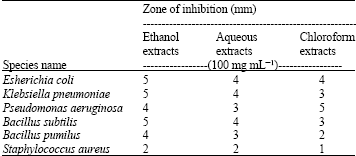
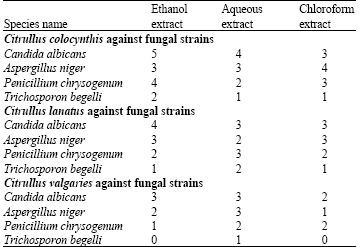
Screening of Anti-microbial Effect in Watermelon (Citrullus sp.)
Centre of Advanced Study in Marine Biology, Faculty of Marine Science,Annamalai University, Parangipettai - 608 502, Tamil Nadu, India
T. Ramanathan
Centre of Advanced Study in Marine Biology, Faculty of Marine Science,Annamalai University, Parangipettai - 608 502, Tamil Nadu, India
N. Ravichandran
Head-Biotechnology Division, Sun Biotech Ltd., 5th Floor Global Chambers,Andheri (W) Mumbai-400053, Maharashtra, India
L. Ramkumar
Centre of Advanced Study in Marine Biology, Faculty of Marine Science,Annamalai University, Parangipettai - 608 502, Tamil Nadu, India