Research Article
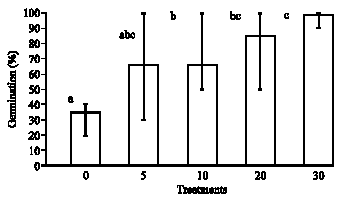
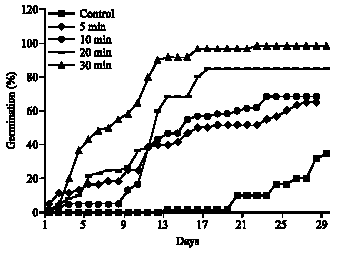
Effects of Scarification Chemical Treatments on the Germination of Crotalaria retusa L. Seeds
Centro de Investigacion de Ciencias Ambientales, Universidad Autonoma del Carmen calle 56 No. 4 Carmen, Campeche, Mexico C.P. 24180, Mexico
L. Aguilar-Marin
Centro de Investigacion de Ciencias Ambientales, Universidad Autonoma del Carmen calle 56 No. 4 Carmen, Campeche, Mexico C.P. 24180, Mexico
N. De la Cruz-Landero
Centro de Investigacion de Ciencias Ambientales, Universidad Autonoma del Carmen calle 56 No. 4 Carmen, Campeche, Mexico C.P. 24180, Mexico
J.J. Guerra-Santos
Centro de Investigacion de Ciencias Ambientales, Universidad Autonoma del Carmen calle 56 No. 4 Carmen, Campeche, Mexico C.P. 24180, Mexico
R. Brito
Centro de Investigacion de Ciencias Ambientales, Universidad Autonoma del Carmen calle 56 No. 4 Carmen, Campeche, Mexico C.P. 24180, Mexico
E. Guevara
Centro de Investigacion de Ciencias Ambientales, Universidad Autonoma del Carmen calle 56 No. 4 Carmen, Campeche, Mexico C.P. 24180, Mexico
R. Gelabert
Centro de Investigacion de Ciencias Ambientales, Universidad Autonoma del Carmen calle 56 No. 4 Carmen, Campeche, Mexico C.P. 24180, Mexico