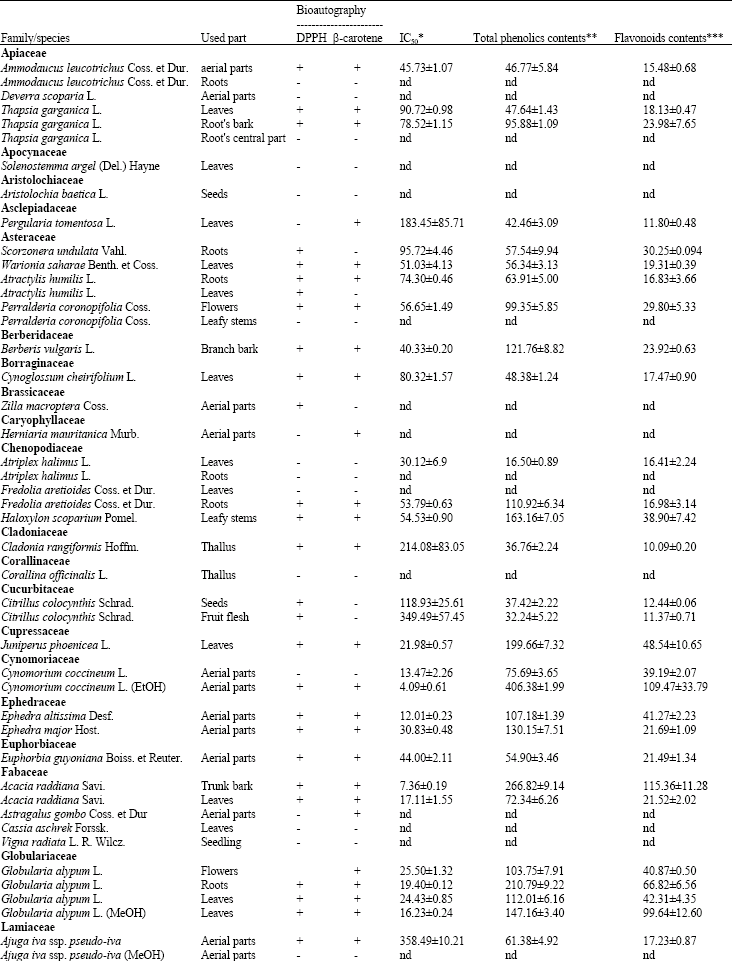
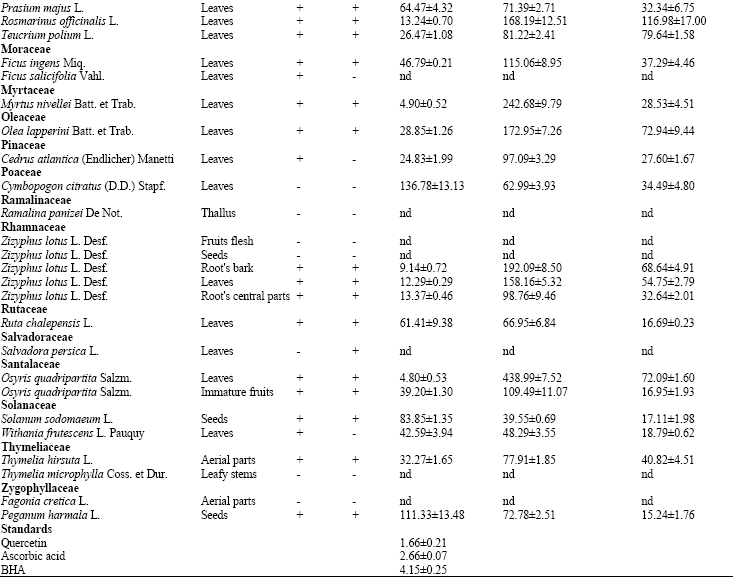
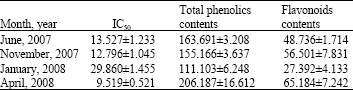
Research Article
Screening of the Antioxidant Potential of Some Algerian Indigenous Plants
Laboratoire de Biochimie Vegetale et des Substances Naturelles, Departement de Biologie,Faculte des Sciences, Universite d`Oran, BP 1524, 31000 Oran El-Menouar, Algeria
H. Benamar
Laboratoire de Biochimie Vegetale et des Substances Naturelles, Departement de Biologie,Faculte des Sciences, Universite d`Oran, BP 1524, 31000 Oran El-Menouar, Algeria
M. Bennaceur
Laboratoire de Biochimie Vegetale et des Substances Naturelles, Departement de Biologie,Faculte des Sciences, Universite d`Oran, BP 1524, 31000 Oran El-Menouar, Algeria
A. Marouf
Laboratoire de Biochimie Vegetale et des Substances Naturelles, Departement de Biologie,Faculte des Sciences, Universite d`Oran, BP 1524, 31000 Oran El-Menouar, Algeria