Research Article
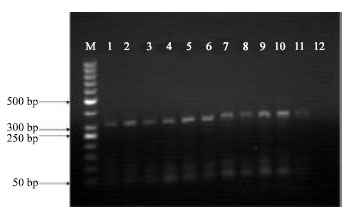
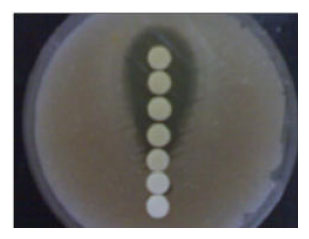
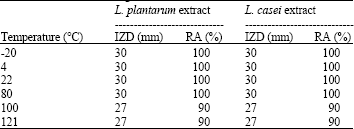
Isolation and Identification of Lactobacillus casei and Lactobacillus plantarum from Plants by PCR and Detection of their Antibacterial Activity
Department of Microbiology, School of Medicine, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran
M. Jorfi
Department of Microbiology, School of Medicine, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran
A.D. Khosravi
Department of Microbiology, School of Medicine, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran
A.R. Samarbafzadeh
Department of Microbiology, School of Medicine, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran
A. Farajzadeh Sheikh
Department of Microbiology, School of Medicine, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran