ABSTRACT
This study was undertaken to investigate the putative protective effect with antioxidant potential of the oyster mushroom (Pleurotus ostreatus) in glycerol-induced Acute Renal Failure (ARF) in rats. Four groups of rats were employed in this study, group I served as control, group II was given a single intramuscular dose of 50% glycerol in olive oil (8 mL kg-1 b.wt.), group III was given mushroom in the diet (62.5 g kg-1) and in drinking water (2.5 g L-1) for 3 weeks and group IV was given mushroom as the third group then was given a single intramuscular dose of 50% glycerol in the day 22 of mushroom administration. Oxidative stress markers in kidney, kidney abnormalities as well as hematological alteration were estimated. Glycerol treatment resulted in a marked renal oxidative stress, a significantly deranged renal function and a reduction in all hematological tested parameters. Pre-treatment with mushroom markedly reduced elevated thiobarbituric acid reacting substances, reduced glutathione, protein carbonyl and resorted the depleted renal antioxidant enzymes, attenuated renal dysfunction and the disturbances observed in hematological and biochemical parameters. These results suggest that Pleurotus ostreatus may have ability to protect the renal damage involved in acute renal failure in rats.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/jbs.2009.746.752
URL: https://scialert.net/abstract/?doi=jbs.2009.746.752
INTRODUCTION
Acute Renal Failure (ARF) is a syndrome characterized by an acute loss of renal function. Yahuda (1993) reported that the acute volume depletion models of glycerol-induced ARF are more closely related to the syndrome of ARF in humans than is the chronic dehydration model. Glycerol induced myoglobin ARF shows many of the hall marks of the crush syndrome, the archetypical form of human ARF that first led to widespread recognition of this disorder in man (Allen and Donald, 1989). Also, glycerol promotes free radical formation lipid peroxidation and renal dysfunction (Halliwell and Gutteridge, 1990).
Lipid peroxidation, a process induced by free radicals, leads to oxidative deterioration of polyunsaturated lipids under normal physiological conditions only low levels of lipids occur in the body tissues. The excessive generation of free radicals leads to peroxidative changes that ultimately result in enhanced lipid peroxidation (Rikans and Hornbrook, 1997). Melondialdehyde (MDA), a secondary product of lipid peroxidation, is used as an indicator of tissue damage (Ohkawa et al., 1979).
Mushroom is an important source of nutrients and of physiologically beneficial, non-toxic medicine (Wasser and Weis, 1999). In fact, mushroom has been used in folk medicine throughout the world since ancient time, these are reported to contain large amounts of vitamin A, C and β-carotene all of which have protective effects because of their antioxidant properties (Antonia et al., 2002).
Reactive oxygen species are believed to be usually generated in aerobic cells, aerobic organisms are provided with antioxidant defense systems that could avert damage due to oxidative stress (Sies, 1985). The major antioxidant defense systems are composed of antioxidant enzymes, including superoxide dismutase (SOD) and catalase (CAT) and biological antioxidants, including reduced glutathione (GSH) (Klivenyi et al., 2000).
Supplementation of antioxidants could conceivably protect the human body from free radicals and retard the progress of many chronic diseases as well as lipid peroxidation (Gülçin et al., 2003).
Mushrooms contain many phenols which are very efficient scavengers of peroxy radicals (Antonia et al., 2002). The extract of the mushrooms has been reported to remove the hyperoxide radical, the main factor in the human ageing process (Liu et al., 1997).
Many medical properties have attributed to Mushrooms (Borchers et al., 1999), including inhibition of platelets aggregation (Hokana and Hokana, 1981), reduction of blood cholesterol concentrations (Aletor, 1995), prevention or alleviation of heart disease and reduction of blood glucose level (Manzi and Pizzoferrato, 2000). Mushrooms have also been reported to block induced liver lipid peroxidation (Lin et al., 1998).
Since, study has tended to focus on the dietary value of edible mushrooms and their possible use to treat oxidative stress.
In the present study, we investigate the protective effect of Oyster mushroom (Pleurotus ostreatus) and its possible use to decrease oxidative stress in glycerol-induced acute renal failure in rats.
MATERIALS AND METHODS
Materials (Experimental animals): Adult male albino rats weighing 100-120 g were used in this study. They were placed in separate cages and allawed food and water ad libitum. They were kept under suitable air flow and temperature during the whole period of experimentation.
Plant materials: Oyster mushroom (Pleurotus ostreatus) obtained from the local market. The fruiting parts of this plant was air dried in an oven at 30-35°C, powdered with an electric grinder and stored at room temperature (20± 2°C). An aqueous extract of mushroom were prepared by immersion the powdered material at 2.5 g L-1 in boiling distilled water and infused for 15 min, the suspension was filtered and stored at -20°C for consumption as drinking fluid.
Methods (Animal grouping): Animals were divided into four groups of five rats each as follows:
| • | Group I: Rats served as control receiving no treatment |
| • | Group II: Rats received a single intramuscular injection of 50% (8 mL kg-1 b.wt.) glycerol according to (Yahuda, 1993) |
| • | Group III: Rats received daily the mushroom for 3 weeks (dried homogenated mushroom incorporated into diet (62.5 g kg-1) and drinking water (2.5 g L-1) according to Swanston-Flatt et al. (1989) and Gray and Flatt (1998) |
| • | Group IV: Rats received daily mushroom for 3 weeks as group III then they received a single intramuscular injection of 50% (8 mL kg-1 b.wt.) glycerol in the day 22 |
At the end of the experimentation period, animals were housed in metabolic cages overnight, where urine samples were collected, centrifuged and stored at -20°C for biochemical analysis.
Fasted animals were sacrificed at the designated times, blood samples were collected in non heparinized glass centrifuge tubes and centrifuged for 15 min at 1000 x g, sera were separated and stored in deep freezer till further biochemical analysis. Another heparinized blood sample used for the determination of some hematological parameters, kidney was taken after the dissection of the animals, homogenized and stored at deep frozen for biochemical analysis.
Biochemical analysis: Lipid peroxidation TBARS was assessed as (Ohkawa et al., 1979), protein carbonyl was determined as (Smith et al., 1991), reduced glutathione (GSH) and superoxide dismutase (SOD) activities was determined by the method of Nishikimi et al. ( 1972).
Alkaline phosphatase was determined according to Belfield and Goldberg (1971), total protein was assessed as Henry (1964), creatinine was measured by the method of Henry (1974), urea was estimated by the method of Patton and Grouch (1977), serum and urine minerals (Na+, K+, Ca++ and Zn) were estimated by an atomic absorption spectrophotometer (Zettner and Seligson, 1964).
Hematological studies: Red Blood Corpuscles (RBCs), White Blood Cells (WBCs), blood platelets (PLTs) count, hemoglobin content (Hb), hematocrit percent (HCt %), Mean Corpuscular Volume (MCV) and Mean Corpuscular Hemoglobin (MCH) were determined using a Sysmex cell counter (Sysmex, Japan) (Dacie and Lewis, 1991).
Statistical analysis: The obtained results were evaluated by one way ANOVA test and post comparison was carried out with Tukey test. The values of p≤0.05 were considered statistically significant (Snedecor and Cochran, 1989).
RESULTS
Table 1 showed the effect of glycerol and mushroom administration on the activities of renal MDA, PC, GSH and SOD. The data revealed that, mushroom administration daily for 3 weeks did not induce significant changes in the tested parameters as compared with those of control. An oxidative stress resulted by a significant increase in MDA and PC and significant decrease in GSH and SOD activities in glycerol treated group as compared to the corresponding values of the control group. An improvement was recorded for these parameters, especially GSH activity in the mushroom and glycerol administered group.
Kidney functions tests were shown in Table 2, a significant decrease in total serum protein content, while a significant increase in both serum and urine creatinine and urea after the intramuscular injection of glycerol were demonstrated. In addition a significant increase in ALPase in the serum and urine in glycerol treated rats and a significant increase in serum K+ and Ca++ levels but Na+ and Zn were decreased while the reverse occurred in urine mineral contents.
| Table 1: | Renal MDA, PC, GSH and SOD in control and different treated groups |
 | |
| Data are expressed as Mean±SD of 5 animals. aSignificant difference as compared to control group and bSignificant difference as compared to glycerol induced animal group according to Tukey test | |
| Table 2: | Some biochemical parameters in serum and urine in control and different treated groups |
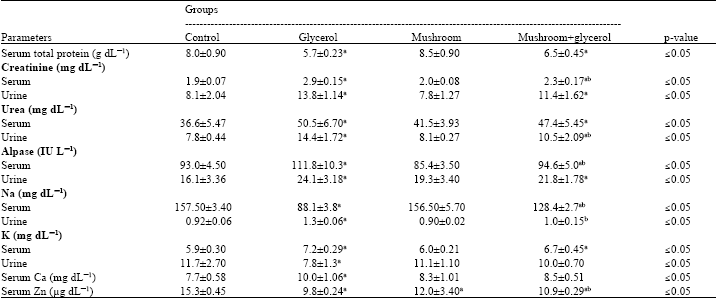 | |
| Data are expressed as Mean±SD of 5 animals. aSignificant difference as compared to control group and bSignificant difference as compared to glycerol induced animal group according to Tukey test | |
| Table 3: | Hematological parameters of control and different treated groups |
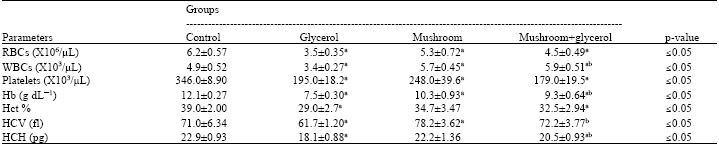 | |
| Data are expressed as Mean±SD of 5 animals. aSignificant difference as compared to control group and bSignificant difference as compared to glycerol induced animal group according to Tukey test | |
On the other hand, Mushroom treatment for three weeks prior to glycerol injection caused slight amelioration in all parameters included in Table 2.
Hematological data showed RBCs, WBCs, platelets counts, Hb, Hct%, MCV and MCH values in control group and different treated groups in Table 3. A significant decrease in all of the above mentioned parameters were recorded in glycerol treated rats. The administration of mushroom preglycerol injection resulted in a pronounced improvement in most of them.
DISCUSSION
Mushroom is an important source of nutrients and of physiologically beneficial, non-toxic medicine (Wasser and Weis, 1999).
Since ancient times, Pleurotus species, cultivated on a commercial scale world-wide since these have been proven to be a good source of almost all essential amino acids, today a great deal of public interest in the use of mushroom as a good antioxidative agent which has a protective effect (Antonia et al., 2002).
The current study demonstrated the protective effect of Oyster mushroom (Pleurotus ostreatus) on the lipid peroxidation and the activities of antioxidant enzymes, the renal functions as well as the hematological parameters against renal failure glycerol induction.
Glycerol treated group showed depletion in antioxidant system which indicated by the significant decrease in the levels of GSH and SOD enzymes and the increase in the oxidative stress, these results are in accordance with Devinder et al. (2004).
Mushroom showed no significant adverse effect for the lipid peroxidation, antioxidant defense system and most of the estimated biochemical parameters. The rats administered with mushroom prior to glycerol injection showed equilibrium between free radicals and antioxidant defense system compared to the rats treated with glycerol only.
The intramuscular administration of hypertonic glycerol induces myolysis and hemolysis affords a faithful and widely utilized model of hemeprotien- induced renal injury (Dubrow and Flamenbaum, 1983; Zager, 1996). The hemeprotein induced renal injury represents the integrated effects of three major pathophysiologic mechanisms: renal vasoconstriction, direct cytotoxicity and cast formation (Dubrow and Flamenbaum, 1988; Zager, 1996). The biochemical finding of renal injury so induced have produced persuasive evidence incriminating oxidative stress as an important mechanism for such injury (Shah and Walker, 1988; Paller, 1988; Baliga et al., 1996). The pigments, hemoglobin and myoglobin themselves, are unlikely to induce acute renal failure, but their presence within the systemic circulation during period of acidosis, dehydration, shock or other conditions associated with reduced renal perfusion may lead to both direct toxic and hemodynamic abnormalities resulting in acute renal failure (Dubrow and Flamenbaum, 1983). In agreement with these results (Yoon et al., 2008) recorded that glycerol injection significantly increased the serum MDA in kidney, moreover, SOD was markedly reduced.
Glutathione is a sugar, non-protein thiol in living organisms which performs a key role in coordinating innate antioxidant defence mechanisms. The GSH is involved in the maintenance of normal structure and function of cells, probably by its redox and detoxification reactions (Gueeri, 1995). In the present study, level of GSH was found to be lower in glycerol treated group than that of the control one, a result which is in agreement with An and et al. (1996). This lowered GSH level may result from increased utilization of GSH by antioxidant enzymes such glutathione peroxidase (GPX), which scavenge H2O2. Interestingly in the present study, when mushroom extract was administered then glycerol was administered to rats, the GSH level was found to be very similar to that in control group. This strongly suggests the antioxidant potential of the extract of the used mushroom.. These results are in agreement with Jayakumar et al. (2007) reported that administration of mushroom extract markedly elevated the level of SOD activity indicating the antioxidant potential of the mushroom P. ostreatus. Living tissues are endowed with innate antioxidant defense mechanisms, including superoxide dismutase (SOD) which catalyzes the removal of superoxide radical, which would otherwise damage the membrane and biological structures. A reduction in the activity of this enzyme is associated with the accumulation of highly reactive free radicals, leading to deleterious effects such as a loss of integrity and function of cell membranes (Sheela and Angusti, 1995).
Rats treated with mushroom preglycerol, can attenuate the changes in lipid peroxidation and oxidative stress, its antioxidant action may be due to its ability to scavenge free radicals and to enhance the cellular antioxidant like reduced glutathione and super oxide dismutase and thereby it prevents lipid peroxidation (Baiba et al., 2002), a result which is in agreement with Nayana and Janardhanan (2000), who reported that the ethyl acetate and methanol extracts of Pleurotus florida exhibit potent hydroxyl radical scavenging and lipid peroxidation inhibition activities and may be due to its high essential amino acids contents as reported by Mattila et al. (2002). In addition, P. ostreatus has been reported to possess excellent reducing power of ferric ions (Lin, 1999).
Mushroom has been proved to be a good source of many mineral elements such as K, P, Zn, Cu and Se (Mattila et al., 2002). Stijve (1977) stated that the highest selenium concentration are found in almost all popular edible mushroom, so the physiological protective capacity of the used mushroom in this study can be referred to its selenium content. Selenium is an integral part of the selenoenzyme glutathione peroxidase.
The observed reduction in the MDA level following of the antioxidant activity of P. ostreatus is agreed with Jayakumar et al. (2007). Antioxidants are necessary for preventing the formation of free radicals and they inhibit some of the deleterious actions of reactive oxygen species on lipids, DNA and proteins (Halliwell, 1996).
Impairment in rats renal functions injected with glycerol, especially significant increase in the two important pathological parameters, serum creatinine and urea, accompanied with an elevation in ALPase activity meanwhile a significant decrease in serum total protein was recorded with disturbance in minerals level in both serum and urine. These results are in agreement with (Yokozawa et al., 1996), who reported the alterations of renal parameters in rats injected with glycerol. Also, Kim et al. (2004) recorded that both urea nitrogen and creatinine levels were considerably increased indicating pathological impairment of renal function by glycerol induced ARF. A significant increase in serum creatinine also recorded by Devinder et al. (2004) and Yoon et al. (2008).
The obtained increase in serum creatinine and urea may be also attributed to the reduction in urea clearance and glomerular filtration rate as reported by Heather et al. (2004), in addition, severely damage in proximal tubule cells may lead to the obtained data, a view which agree with Yoon et al. (2008).
The decline in serum total protein and the elevation in urea and creatinine may be attributed to the increase in protein catabolism (Abdel Moneim et al., 1999) superimposed to glomerular or tubular destructive changes (Ravel, 1984). This disturbance may be also attributed to either intense vasoconstriction or renal tubular necrosis, or due to inordinate amount of myoglobin released into the systemic circulation with its prompt discharge into the renal tubules (Better and Stain, 1990).
A significant amelioration in urea and creatinine levels in serum and urine was recorded in mushroom and glycerol administered group, results which may be due to the presence of antioxidant compounds that interfere with inducible nitric oxide synthase activity, in addition to their powerful radical scavengers (Hanasaki et al., 1994) and the attenuation of protein catabolism and rate of gluconeogenesis (Rawi et al., 1998; Abdel Moneim et al., 1999, 2001) and/or diminished activity of urea cycle.
Glycerol induction cause significant increase in serum and urine ALPase, this result may be attributed to the production of free radicals which could have affected the cellular permeability leading to elevation in circulatory level of this enzyme (Amer and Areida, 2005). On the other hand, improvement in ALPase level in group administred mushroom preglycerol injection, a result which may be due to its antioxidative capacity and scavenging free radicals.
Regarding minerals, significant decrease in serum Na+ content and Zn but K+ and Ca++ levels were increased while the reverse occurred in urine, a result which may be due to excretion of Na+ as Kim et al. (2004), recorded an increase in the excretion of sodium after injection with glycerol. The resulted disturbances in minerals content in serum and urine in glycerol administered group may be also due to hypovolemia metabolic disturbances as reported by Zager and Gamelin (1989). Obtained hyperkalemia is in agreement with Singh et al. (2004) and may be attributed to the complications associated with rhabdomyolysis.
Obtained changes in Na+ and K+ content resulted due to cell membrane damage which lead to disturbance in Na+ and K+ pumping and disorders in membrane permeability, a view which in agreement with (Kattab et al., 2003). Administration of mushroom might improved the stability of the cell mechanism so protects the cell against abnormal permeability.
Adverse effects in hematological parameters were noted in rats treated with glycerol. The reduction in erythrocytes, leucocytes and platelets counts with subsequent decline in the values of Hb, Hct%, MCV and MCH, suggested that there is an etiologic relationship between ARF and anemia through different suggested mechanisms such as bone marrow cells destruction and decrease or delay in mitosis (Hall, 2001) or by the increase of erythrocytes osmotic fragility. The disturbances in RBCs count often reflect an imbalance between its production and loss, non regenerative anemia arises from reduced erythrocytes production. Dysfunction of the kidney induced by glycerol may have a negative effect on erythropoiesis that result in a low RBCs production and RBCs count (Hassan et al., 2008).
Concerning the observed decrease in WBCs count may reflect alterations in the immune function. The decrease in platelet count may be due to glycerol inhibiting bone marrow activity or may be due to decreased production or increased consumption of platelet or due to the increased of platelets aggregation (Moncada et al., 1991). Administration of mushroom caused an improvement in most recorded hematological parameters. These ameliorative effects could be due to the lowered lipid peroxidation level in cell membranes leading to decreased susceptibility of RBCs to hemolysis and/or due to the prevention of free radicals induced damage through its antioxidant activity achieved by its various phytochemical contents (Frankel et al., 1998).
In conclusion, it seems that pretreatment of mushroom Pleurotus ostreatus play a beneficial role in the glycerol induced acute renal failure in rats and possesses potential to contract the oxidative stress.
REFERENCES
- Abdel-moneim, A., E. El-zyat and O. Ahmed, 1999. Studies on the effects of glimepiride and some antidiabetic plants on protein metabolism of streptozotocin diabetic rats. J. Egypt. Ger. Soc. Zool., 28: 79-98.
Direct Link - Aletor, V.A., 1995. Compositional studies on edible tropical species of mushrooms. Food Chem., 54: 265-268.
CrossRefDirect Link - Allen, L. and E. Donald, 1989. Glomerular hemodynamics in established glycerol-induced acute renal failure in the rat. J. Clin. Invest., 84: 1967-1973.
Direct Link - Amer, M. and S. Areida, 2005. Protective effect of Liv-Plus. A herbal formulation against hepatotoxicity induced by carbon tetrachloride and thioacetamide in male rats. J. Egypt. Ger. Soc. Zool., 44: 199-220.
Direct Link - Anand, C., V. Anand and R. Agarval, 1996. Antioxidant enzymes, gammaglutamyl transpeptidase and lipid peroxidation in kidney of rats exposed to cigarette smoke. Ind. J. Exp. Biol., 34: 486-488.
Direct Link - Grube, B.J., E.T. Eng, Y.C. Kao, A. Kwon and S. Chen, 2001. White buttom mushroom phytochemicals inhibit aromatase activity and breast cancer cell oroliferation. J. Nutr., 131: 3288-3293.
PubMedDirect Link - Better, O. and J. Stain, 1990. Early management of shock and prophylaxis of acute renal failure in traumatic rhabdmyolysis. New Engl. J. Med., 322: 825-829.
Direct Link - Borchers, H.A., J.S. Stem, R.M. Hackman, C.L. Keen and M.E. Gershwin, 1999. Mushroom tumours and immunity. Proc. Soc. Exp. Med., 221: 281-293.
Direct Link - Devinder, S., C. Vikas and C. Kanwaljit, 2004. Protective effect of naringin , a bioflavonoid on glycerol-induced acute renal failure in rat kidney. Toxicology, 201: 143-151.
Direct Link - Frankel, S., G. Robinson and M. Berenbaum, 1998. Antioxidant capacity and correlated characteristics of 14 unifloral honeys. J. Apic. Res., 37: 27-31.
Direct Link - Gray, A. and P. Flatt, 1998. Insulin releasing and Insulin like activity of Agaricus compestris (Mushroom). J. Endocrinol., 157: 259-266.
Direct Link - Guerri, C. and S. Grisolia, 1980. Influence of prolonged ethanol intake on the levels and turnover of alcohol and aldehyde dehydrogenases and glutathione. Adv. Exp. Med. Biol., 126: 365-384.
CrossRefDirect Link - Gulcin, I., M. Oktay, E. Kirecci and O.I. Kufrevioglu, 2003. Screening of antioxidant and antimicrobial activities of anise (Pimpinella anisum L.) seed extracts. Food Chem., 83: 371-382.
CrossRefDirect Link - Halliwell, B. and J.M.C. Gutteridge, 1990. Role of free radicals and catalytic metal ions in human disease: An overview. Methods Enzymol., 186: 1-85.
CrossRefDirect Link - Halliwell, B., 1996. Antioxidants in human health and disease. Annu. Rev. Nutr., 16: 33-50.
CrossRefPubMedDirect Link - Heather, M., W. David and J. Andrew, 2004. Macrophages and the kidney. Curr. Opin. Nephrol. Hypertens., 13: 285-290.
Direct Link - Jayakumar, T., P. Thomas and P. Geraldine, 2007. Protective effect of an extract of the oyster mushroom, Pleurotus ostreatus, on antioxidant of major organs of aged rats. Exp. Gerontol., 42: 183-191.
Direct Link - Kim, H., T. Yokozawa, T. Nakagawa and S. Sasak, 2004. Protective effect of γ-aminobutyric acid against glycerol-induced acute renal failure in rats. Food Chemical Toxicol., 42: 2009-2014.
Direct Link - Manzi, P. and L. Pizzoferrato, 2000. Beta-glucans in edible mushrooms. Food Chem., 68: 315-318.
CrossRefDirect Link - Mattila, P., P. Salo-Vaananen, K. Konko, H. Aro and T. Jalava, 2002. Basic composition and amino acids content of mushroom cultivated in Finland. J. Agric. Food Chem., 50: 6419-6422.
Direct Link - Moncada, S., R. Palmer and E. Higgs, 1991. Nitric oxide :Physiology, pathophysidogy and pharmacology. Pharmacol. Rev., 43: 109-142.
Direct Link - Nishikimi, M., N.A. Rao and K. Yagi, 1972. The occurrence of superoxide anion in the reaction of reduced phenazine methosulfate and molecular oxygen. Biochem. Biophys. Res. Commun., 46: 849-854.
CrossRefPubMedDirect Link - Paller, M., 1988. Hemoglobin and myoglobin-induced acute renal failure in rats. Am. J. Physiol., 255: 539-544.
Direct Link - Rikans, L.E. and K.R. Hornbrook, 1997. Lipid peroxidation, antioxidant protection and aging. Biochimica Biophysica Acta (BBA)-Mol. Basis Dis., 1362: 116-127.
CrossRefDirect Link - Shah, S. and P. Walker, 1988. Evidence suggesting a role for hydroyl radical in glycerol-indnced acute renal failure. Am. J. Physiol., 255: F438-F443.
Direct Link - Sheela, C.G. and K.T. Angusti, 1995. Antiperoxide effects of S-allyl cystein sulphoxide isolated from Allium sativum Linn. and gugulipid in cholesterol diet fed rats. Indian J. Exp. Biol., 33: 337-341.
Direct Link - Singh, D., V. Chander and K. Chopra, 2004. Protective effect of naringin, a bioflavonoid on Glycerol-induced acute renal failure in rat kidney. Toxicology, 201: 143-151.
CrossRefDirect Link - Swanston-Flatt, S.K., C. Day, P.R. Flatt, B.J. Gould and C.Y. Bailey, 1989. Glycaemic effects of traditional European plant treatments for diabetes. Studies in normal and streptozotocin diabetic mice. Diabetes Res., 10: 69-73.
PubMed - Wasser, S.P. and A.L. Weis, 1999. Medicinal properties of substances occurring in higher basidiomycete mushrooms: Current perspective. Int. J. Med. Mushrooms, 1: 31-62.
Direct Link - Yahuda, Z., 1993. Models of glycerol-induced acute renal failure in rats. J. Basic Clin. Phsiol. Pharmacol., 4: 213-228.
Direct Link - Nayana, J. and K.K. Janardhanan, 2000. Antioxidant and antitumor activity of Pleurotus florida. Curr. Sci., 79: 941-943.
Direct Link - Antonia, M., M. Martinez-Tome, A.M. Jimenez, M. Vera Ana, M. Honrubia and P. Parras, 2002. Antioxidant activity of edible fungi (truffles and mushroom): Losses during industrial processing. J. Food Prot., 65: 1614-1622.
Direct Link - Hanasaki, Y., S. Ogawa and S. Fukui, 1994. The correlation between active oxygens scavenging and antioxidative effects of flavonoids. Free Radical Biol. Med., 16: 845-850.
CrossRefPubMedDirect Link - Smith, C.D., J.M. Carney, P.E. Starke Reed, C.N. Oliver, E.R. Stadtman, R.A. Floyd and W.R. Markesbery, 1991. Excess brain protein oxidation and enzyme dysfunction in normal aging and in Alzheimer disease. Proc. Natl. Acad. Sci. USA., 88: 10540-10543.
PubMedDirect Link - Ohkawa, H., N. Ohishi and K. Yagi, 1979. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem., 95: 351-358.
CrossRefPubMedDirect Link - Patton, C.J. and S.R. Crouch, 1977. Spectrophotometric and kinetics investigation of the Berthelot reaction for the determination of ammonia. Anal. Chem., 49: 464-469.
CrossRefDirect Link








