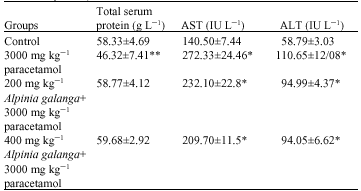
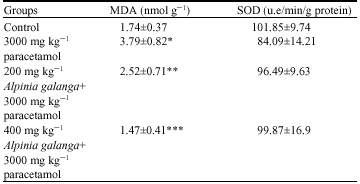
Research Article
Paracetamol Hepatotoxicity in Rats Treated With Crude Extract of Alpinia galanga
Department of Biomedical Sciences, Faculty of Allied Health Sciences, Universiti Kebangsaan Malaysia, Jalan Raja Muda Abdul Aziz, 50300, Kuala Lumpur, Malaysia
Siti Balkis Budin
Department of Biomedical Sciences, Faculty of Allied Health Sciences, Universiti Kebangsaan Malaysia, Jalan Raja Muda Abdul Aziz, 50300, Kuala Lumpur, Malaysia
Victor Feizal
Department of Biomedical Sciences, Faculty of Allied Health Sciences, Universiti Kebangsaan Malaysia, Jalan Raja Muda Abdul Aziz, 50300, Kuala Lumpur, Malaysia