Research Article
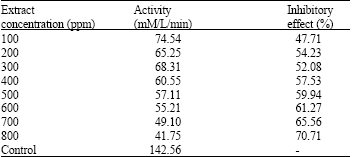
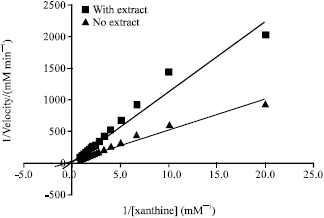
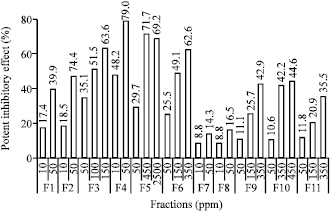
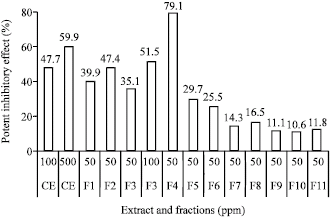
Indonesian Sidaguri (Sida rhombifolia L.) as Antigout and Inhibition Kinetics of Flavonoids Crude Extract on the Activity of Xanthine Oxidase
Biopharmaca Research Center, Bogor Agricultural University, JL. Taman Kencana No. 3, Bogor, 16151, Indonesia
L.K. Darusman
Department of Chemistry, Faculty of Mathematics and Natural Sciences, Bogor Agricultural University, Bogor, 16680, Indonesia
R. Hidayat
Department of Biochemistry, Faculty of Mathematics and Natural Sciences, Bogor Agricultural University, Bogor, 16680, Indonesia