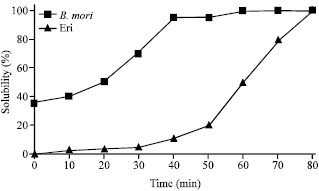
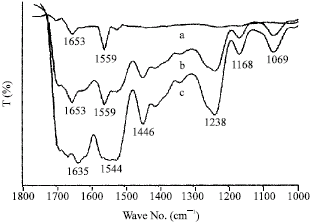
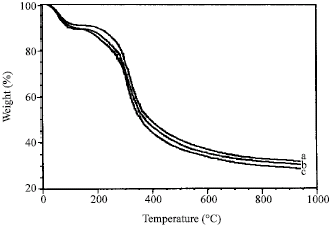
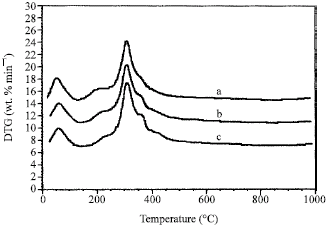
Research Article
Characterization on Eri (Philosamia ricini) Fibroin Film: Morphology, Structure and Thermal Properties
Department of Chemistry, Faculty of Science, Mahasarakham, University, 44150, Thailand
N. Narkkong
Central Instrument Unit, Faculty of Science, Mahasarakham, University, 44150, Thailand
P. Srihanam
Department of Chemistry, Faculty of Science, Mahasarakham, University, 44150, Thailand