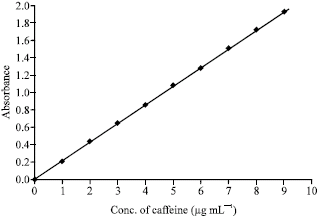
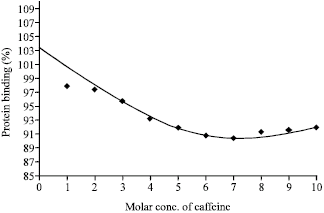
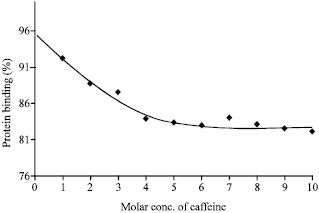
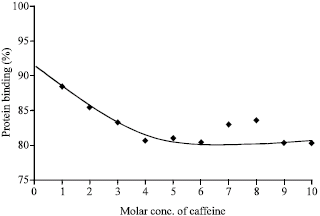
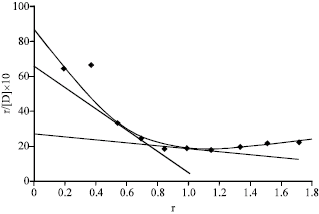
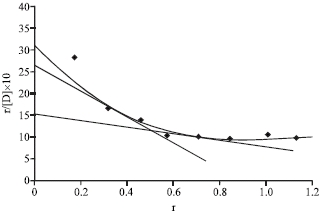
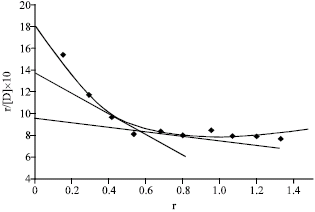
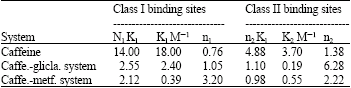
Research Article
In vitro Effects of Gliclazide and Metformin on the Protein Binding of Caffeine in the Aqueous Media
Department of Pharmaceutical Chemistry, Faculty of Pharmacy, University of Dhaka, Dhaka-1000, Bangladesh
A.T.M. Zafrul Azam
Department of Pharmaceutical Chemistry, Faculty of Pharmacy, University of Dhaka, Dhaka-1000, Bangladesh
Md. Shah Amran
Department of Pharmaceutical Chemistry, Faculty of Pharmacy, University of Dhaka, Dhaka-1000, Bangladesh
Md. Amjad Hossain
Department of Pharmaceutical Chemistry, Faculty of Pharmacy, University of Dhaka, Dhaka-1000, Bangladesh