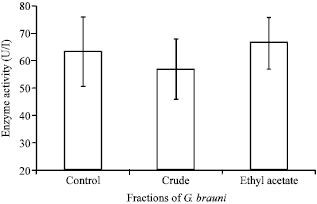
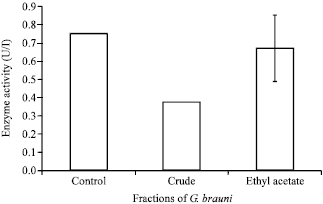
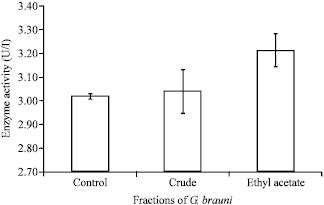
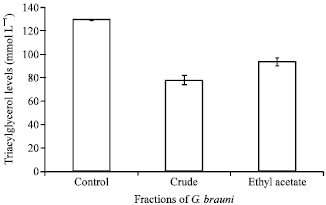
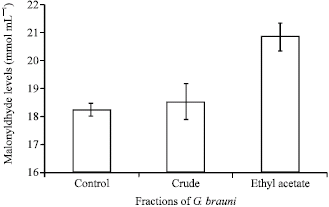
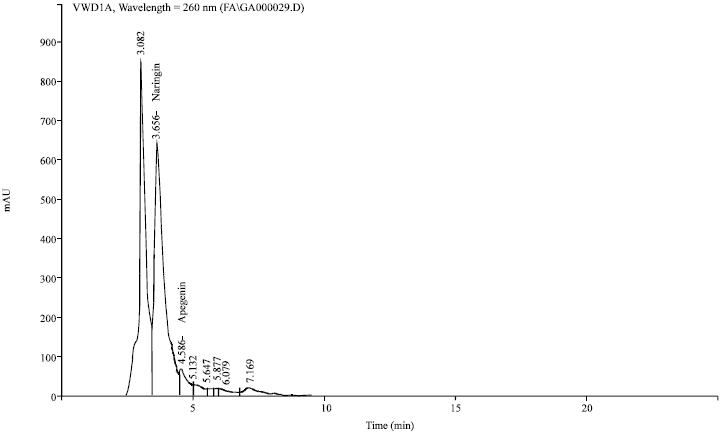
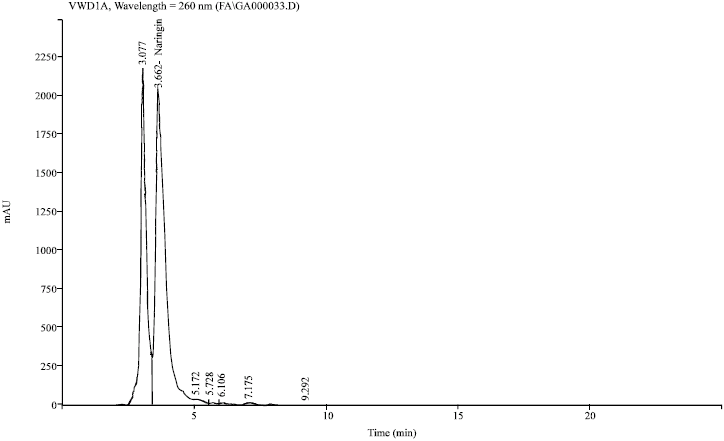
Research Article
Antioxidative Properties of Ethyl Acetate Fraction of Globimetula braunii in Normal Albino Rats
Department of Cell Biology and Genetics, University of Lagos, Akoka-Yaba, Lagos, Nigeria
H. Ogbunugafor
Department of Applied Biochemistry, Nnamdi Azikiwe University, Awka, Anambra State, Nigeria
G.K. Kareem
Department of Cell Biology and Genetics, University of Lagos, Akoka-Yaba, Lagos, Nigeria