Research Article
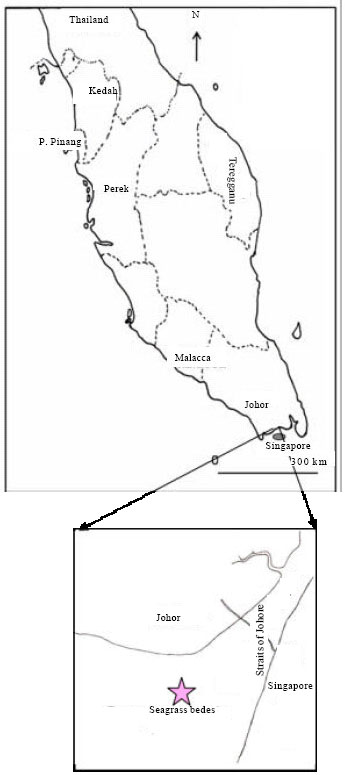
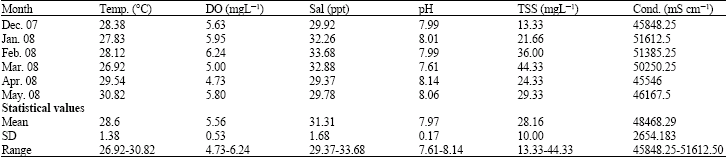
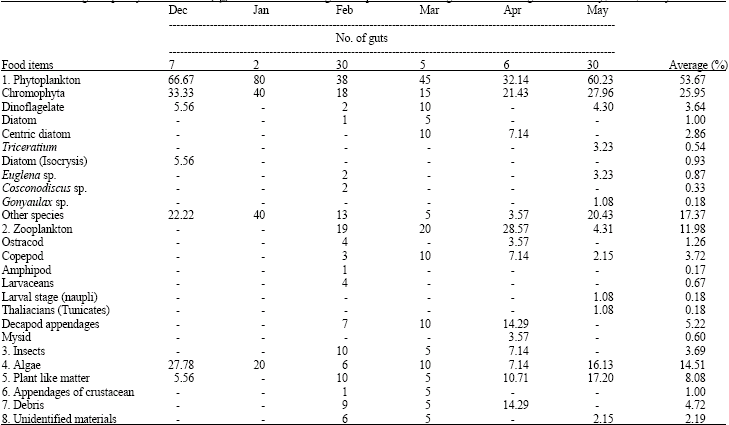
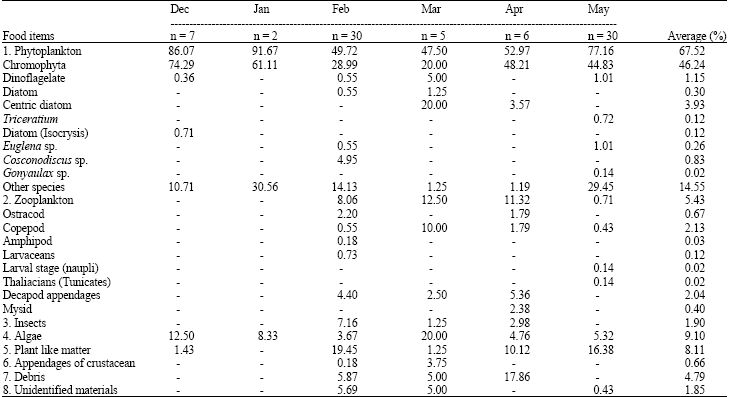
Feeding Habits and Temporal Variation of Diet Composition of Fish Larvae (Osteichthyes: Sparidae) in the Sungai Pulai Seagrass Bed, Johore, Peninsular Malaysia
Laboratory of Marine Science and Aquaculture, Institute of Bioscience, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
A. Arshad
Laboratory of Marine Science and Aquaculture, Institute of Bioscience, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
N. Amrullah
Department of Biology, Faculty of Science, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
S.M. Nurul Amin
Laboratory of Marine Science and Aquaculture, Institute of Bioscience, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
S.K. Daud
Department of Biology, Faculty of Science, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
A.A. Nor Azwady
Department of Biology, Faculty of Science, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
A.G. Mazlan
School of Environmental and Natural Resource Sciences, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, 43600 Bangi, Selangor, Malaysia